
At high pressure, the compressibility factor 'Z' is equal toa
4.8 (517) In stock

4.8 (517) In stock

The compressibility factor a real gas high pressure is:1+ dfrac{RT

4.2: Real Gases (Deviations From Ideal Behavior) - Chemistry LibreTexts

If assertion is true but reason is false.

Gas Laws Compressibility Factor High Pressure & Low Pressure

What is compressibility factor? What is its value for ideal gas

Compressibility Factor - an overview
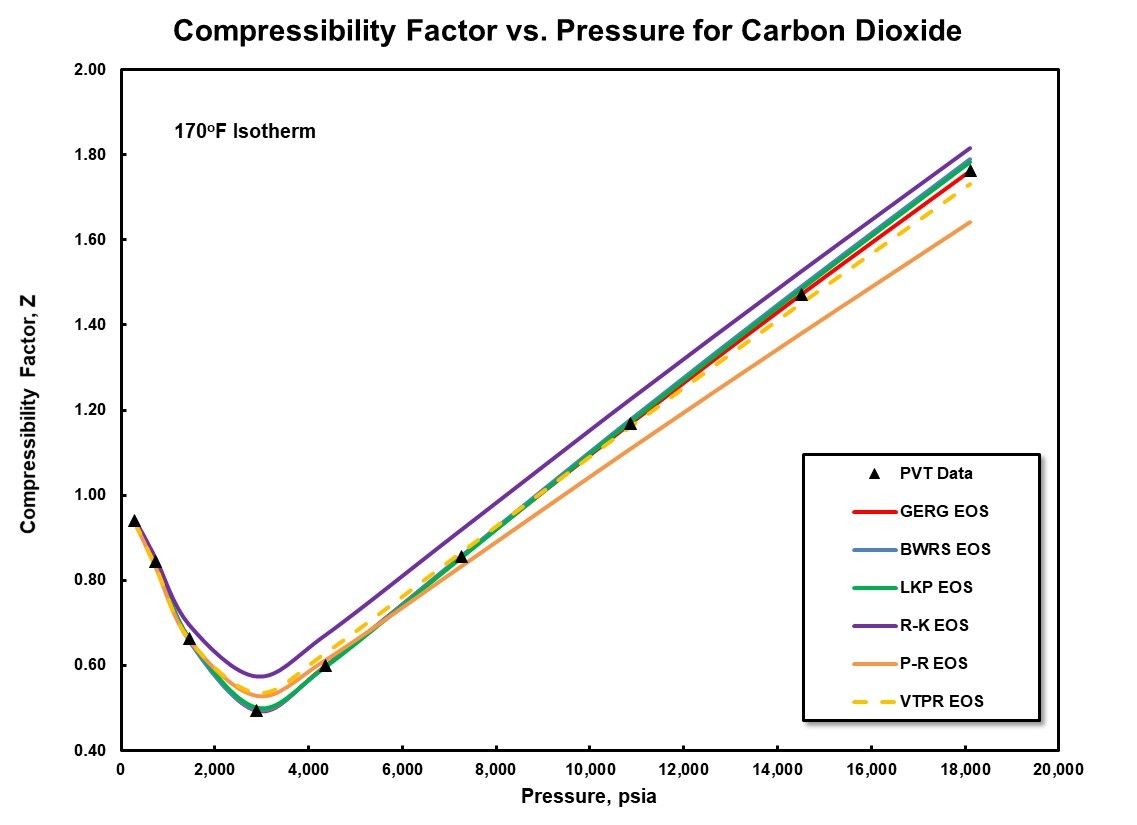
Compressor performance and thermodynamics
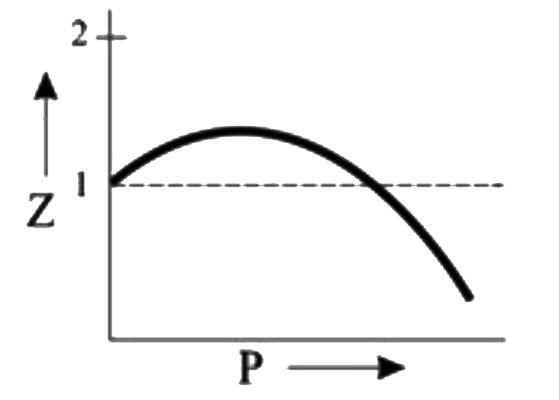
Which of the following represents a plot of compressibility factor (Z)
At very high pressure, the compressibility factor of one mole of a gas

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be
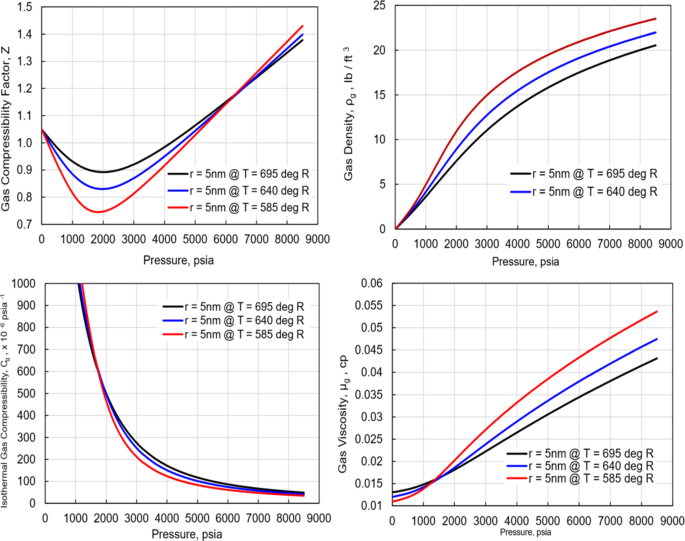
Investigation of the Properties of Hydrocarbon Natural Gases Under Confinement in Tight Reservoirs Due to Critical Properties Shift