Solved] Why is the compressibility factor less than 1 at most conditions?
4.7 (639) In stock
4.7 (639) In stock

Non-ideal behavior of gases (article)
What does a compressibility factor >1 signify, apart from a deviation from the ideal gas behaviour? Is it more compressible? - Quora

Compressibility Factor - an overview

The compressibility factor Z of a gas is less than unity at STP. Therefore

The compressibility factor of a gas is less than 1 at STP. Its molar volume Vm will be
3.1 Ideal gas and ideal gas equation of state – Introduction to Engineering Thermodynamics
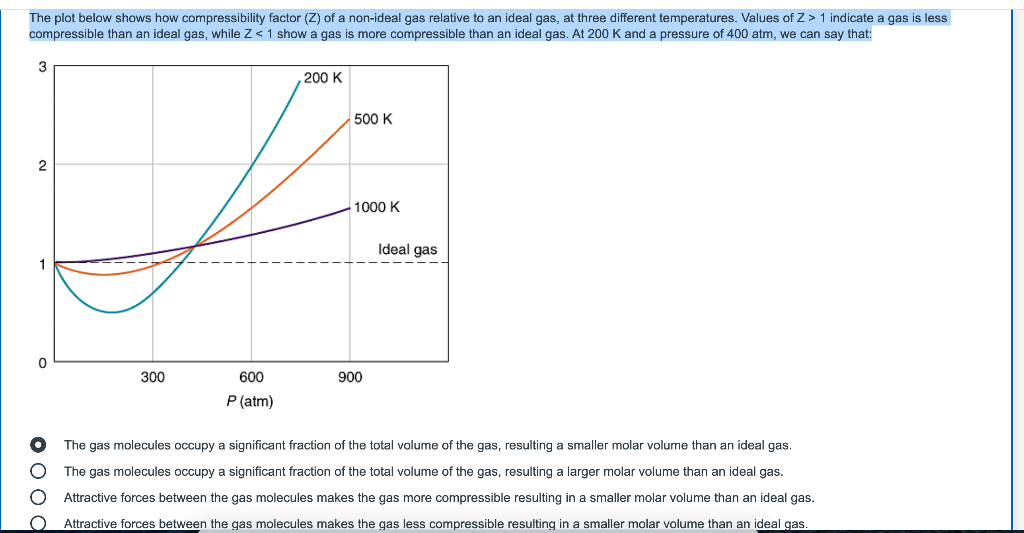
Solved 5/10 5 оооо 1 point Gases confined in small volumes

Solved We begin by showing that the compressibility factor

Ideal Gas Law Equation Compressibility Of Natural Gas - Chemistry

Non-Ideal Gas Behavior Chemistry: Atoms First
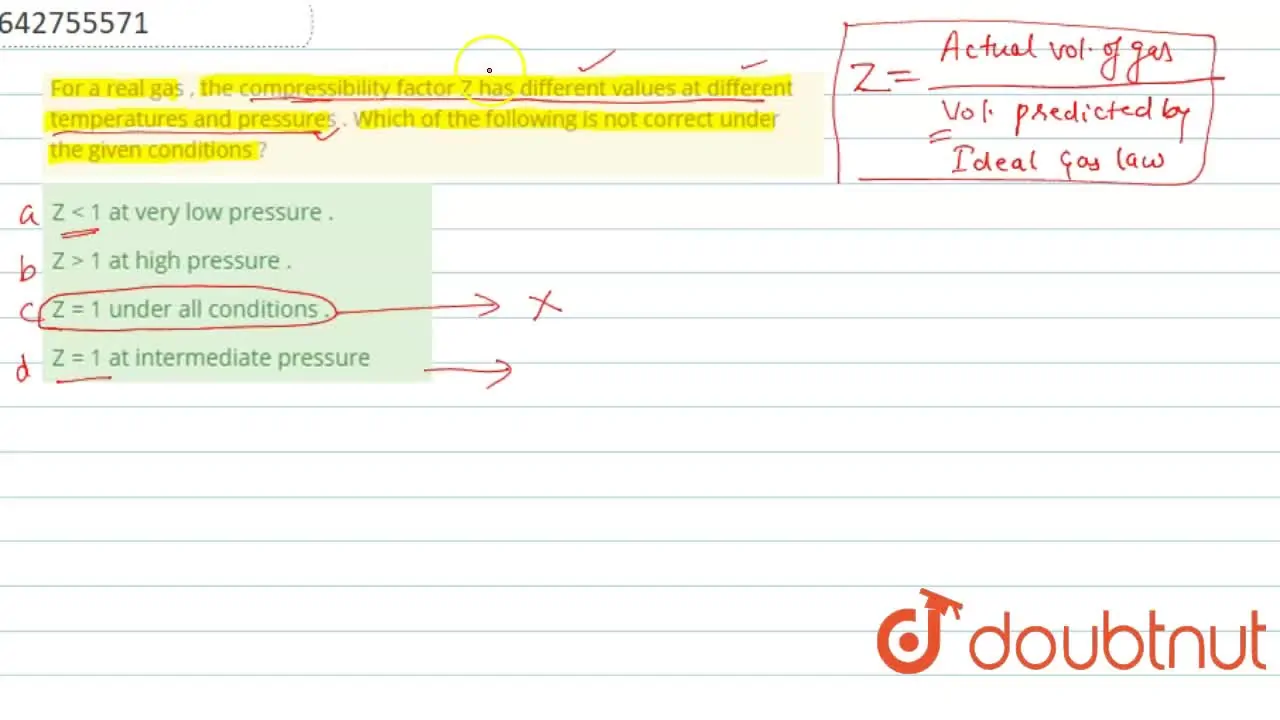
For a real gas , the compressibility factor Z has different values at