
For H(2) gas, the compressibility factor,Z = PV //n RT is
4.6 (735) In stock

4.6 (735) In stock
For H(2) gas, the compressibility factor,Z = PV //n RT is

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas

For an ideal gas, the value of compressibility factor `Z(=(pVm

SOLUTION: State of matter gases liquids and solids - Studypool

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

For H_(2) gas, the compressibility factor,Z = PV //n RT is -, CLASS 12, GASEOUS STATE
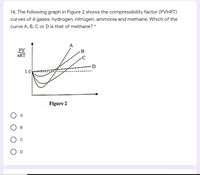
Answered: 14, The following graph in Figure 2…

D) P V- (C) PV- nRT Compressibility factor H2 behaving as real gas is : D) Pb RTV (A) 1 RTV (1-a)