
Write the expression for the compressibility factor (Z) for one mole of a gas. Write the value of Z for an
4.6 (271) In stock

4.6 (271) In stock

Compressibility Factor Z Important Concepts and Tips for JEE Main

SOLVED: Derive the mathematical expression expressing the compressibility factor Z of a real gas depending on the reduced variables; Explain in detail how the volume of the actual gas at a given

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be

Gas Compressibility - an overview

physical chemistry - Pressure vs volume plot for real gas and ideal gas - Chemistry Stack Exchange
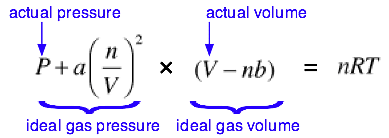
Derivation of Van Der Waals Equation: Real & One Mole of Gas

SOLVED: Derive the mathematical expression expressing the compressibility factor Z of a real gas depending on the reduced variables; Explain in detail how the volume of the actual gas at a given

Compressibility Factor Z

SOLVED: Derive the mathematical expression expressing the compressibility factor Z of a real gas depending on the reduced variables; Explain in detail how the volume of the actual gas at a given

Real-gas z-factor, as attributed to Standing and Katz, 9 plotted as a

Write the expression for compressibility factor (z) for 1 mole of the gas..
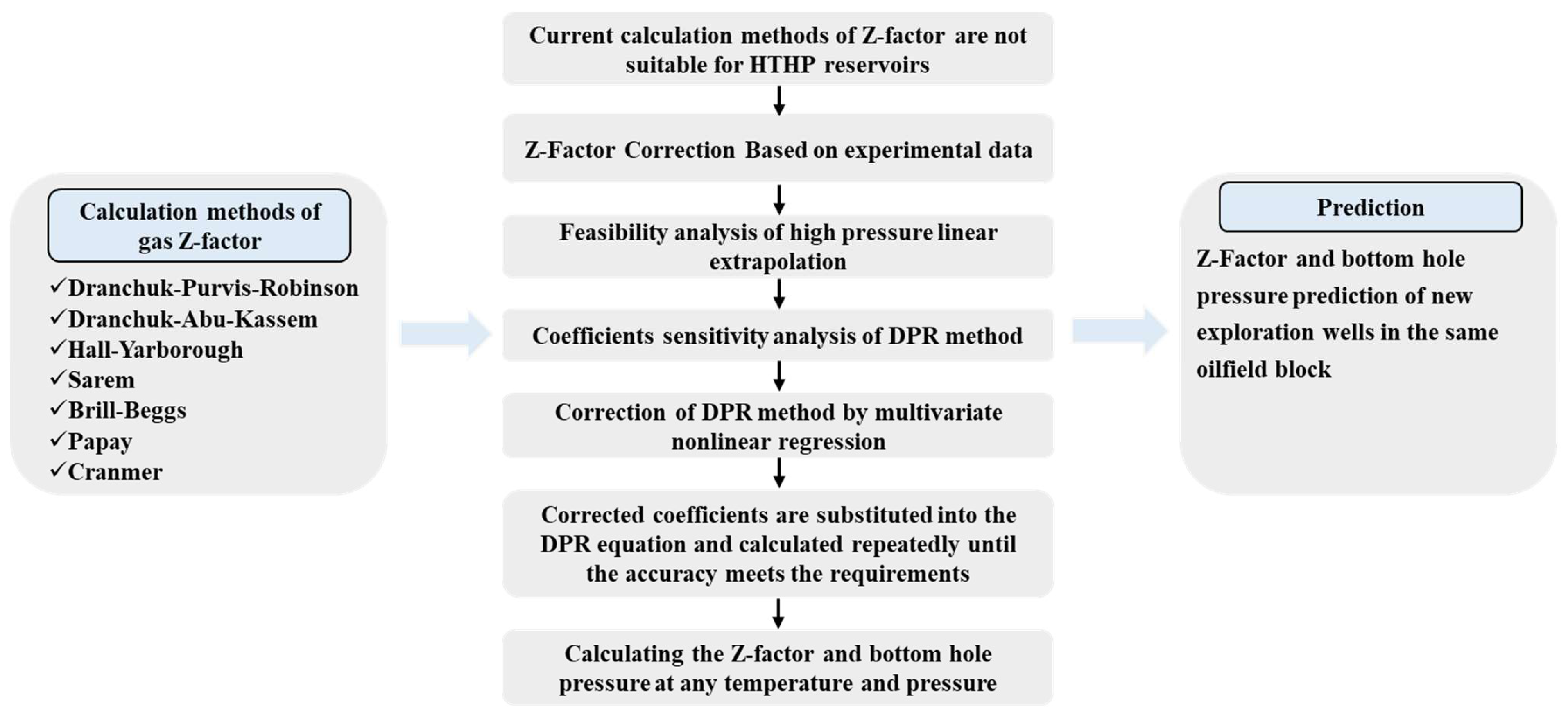
Energies, Free Full-Text

Solved We begin by showing that the compressibility factor