
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
4.7 (189) In stock

4.7 (189) In stock
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state
Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora

Compressibility factor - Wikipedia
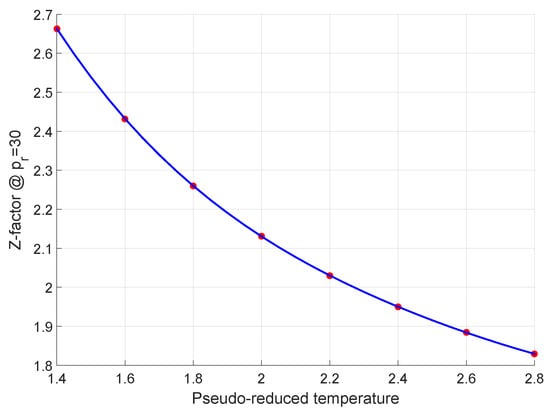
Energies, Free Full-Text

P k nag solution by Shaikh Mohd Aslam - Issuu
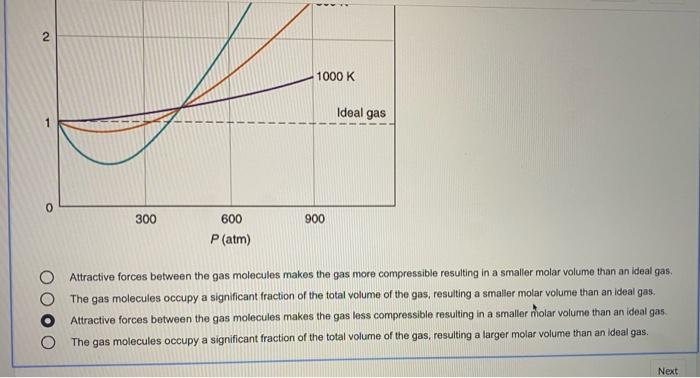
Solved 6 1 point The plot below shows how compressibility
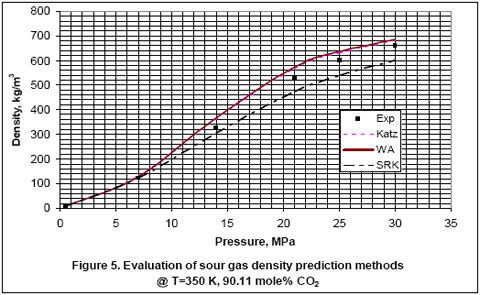
How good are the shortcut methods for sour gas density

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

Determine Compressibility of Gases

e Compressibility factor (Z) for hydrogen WRT pressure and temperature

47. In the following compressibility factor (Z) vs pressure graph 300 K, the compressibility factor of CH4 pressures < 200 bar deviates from ideal behavior because

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

The graph of compressibility factor (Z) vs. P for one mole of a real gas is shown in following
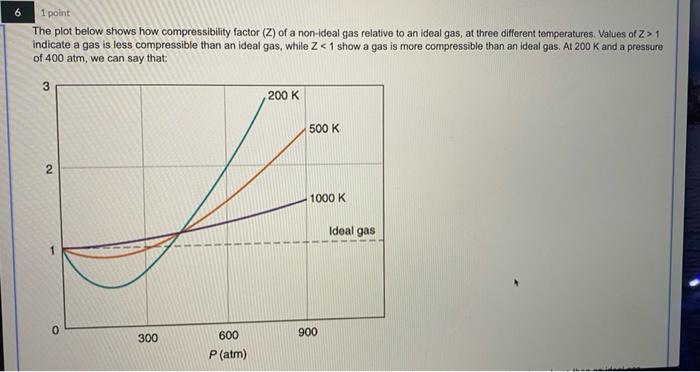
Solved 6 1 point The plot below shows how compressibility