The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT - Sarthaks eConnect
4.6 (226) In stock
4.6 (226) In stock
The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT

A : At high pressure , the compressibility factor Z is (1 + (pb)/(RT))

At low pressures the compressibility factor for a van

Solved We begin by showing that the compressibility factor

Gas compressibility factor Z: Ideal gas vs Real gas
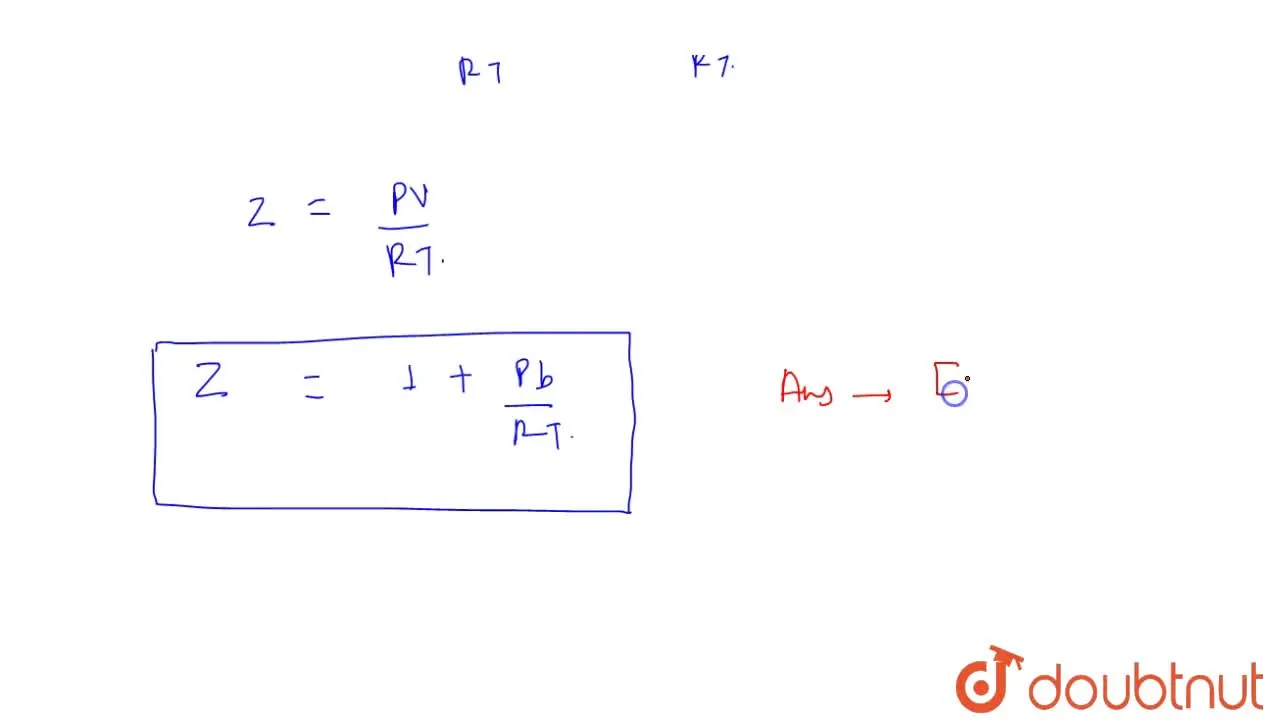
The compressibility factor for a real gas at high pressure is .

The compressiblity factor for a real gas at high pressure is
The given graph represent the variations of compressibility factor

The graph of compressibility factor (Z) vs. P for one mole of a

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

At moderate pressure, the compressibility factor a particular gas

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

Simple Equation Real Gas Compressibility Factor Z

What is compressibility factor? What is its value for ideal gas

SOLVED: The compressibility factor for a real gas at high pressure

3.2 Real gas and compressibility factor – Introduction to