
Indian industry group calls for clearer expiry date labelling rules for topic drugs
4.6 (405) In stock

4.6 (405) In stock
The DCGI needs to clarify expiry date labelling rules for topical drugs according to an Indian trade group, which says some members’ products are being deemed to be ‘misbranded.’

US FDA's Hamburg offers few details on trip to India

3 Million

India must act on drug adulteration – lives around the world are at stake, Dinesh S Thakur and Prashant Reddy Thikkavarapu

17 առօրյա իր, որոնց պիտանելիության ժամկետի մասին մենք նույնիսկ գլխի չենք ընկնում (լուսանկարներ)

Labeling in pharmaceutical packaging

India calls for post-market data on cancer drugs without local trials

Orsl: Devil lies in the details: ORSL sneaks into chemist shelves in a new avatar

Labeling in india
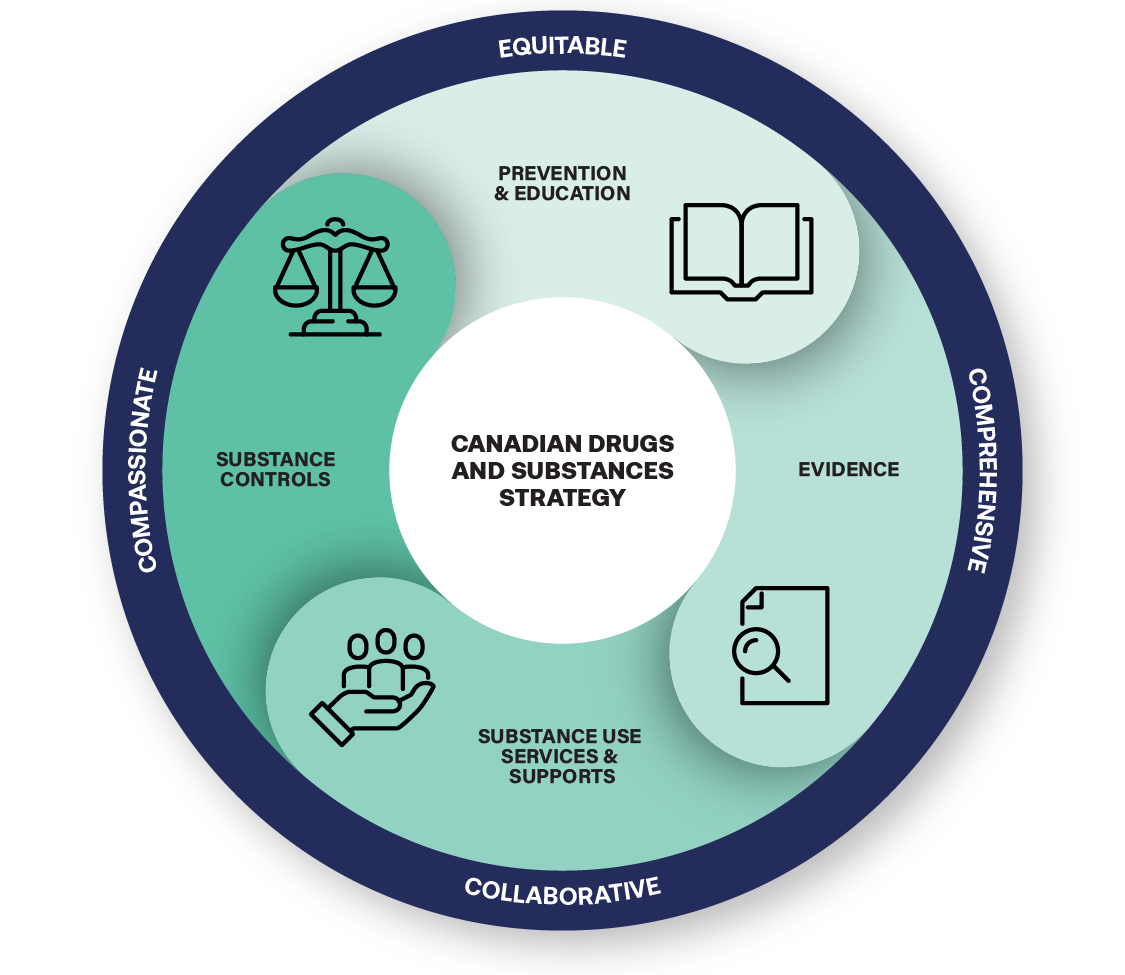
Canadian Drugs and Substances Strategy: Overview