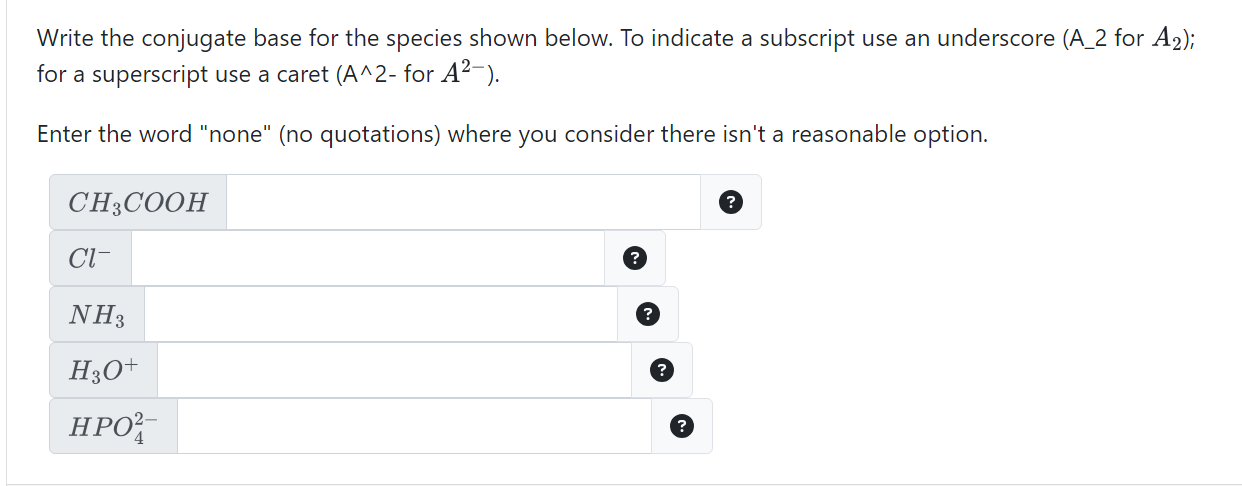
Solved Write the conjugate base for the species shown below.
4.8 (100) In stock

4.8 (100) In stock
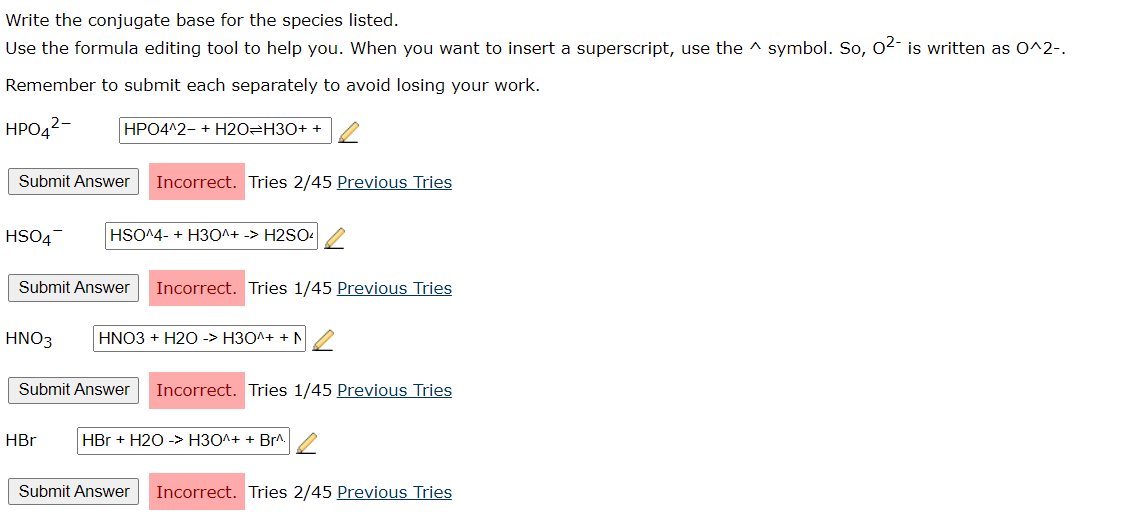
Solved Write the conjugate base for the species listed. Use

Use your understanding of molecular structure to explain why the conjugate bases of acids like formic acid CHOOH, acetic acid CH3COOH, and phosphoric acid are only stable enough to be weak acids;

Write the formula for the conjugate base of each acid. a. HCl
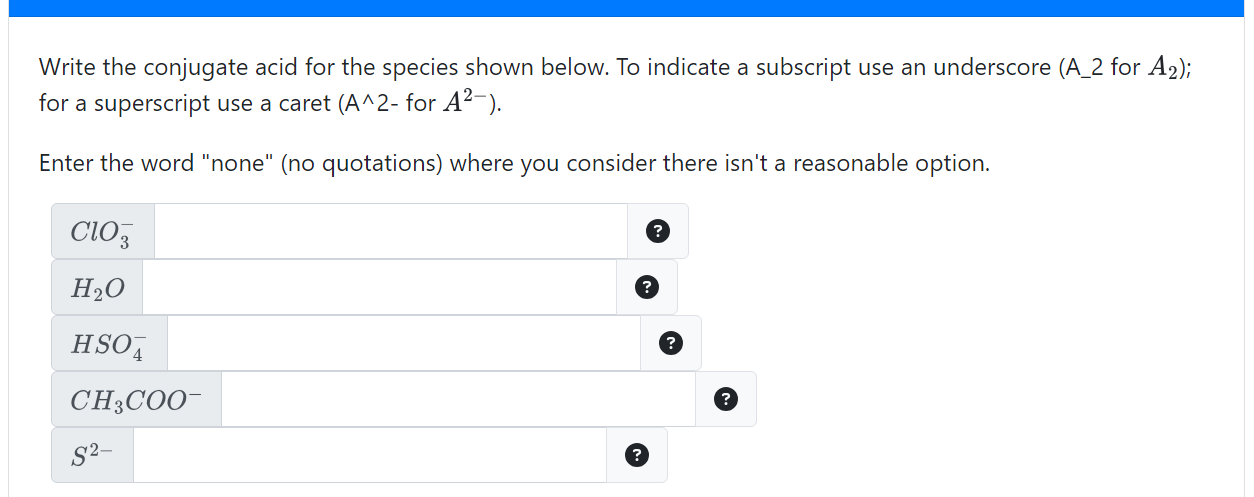
Solved Write the conjugate acid for the species shown below.
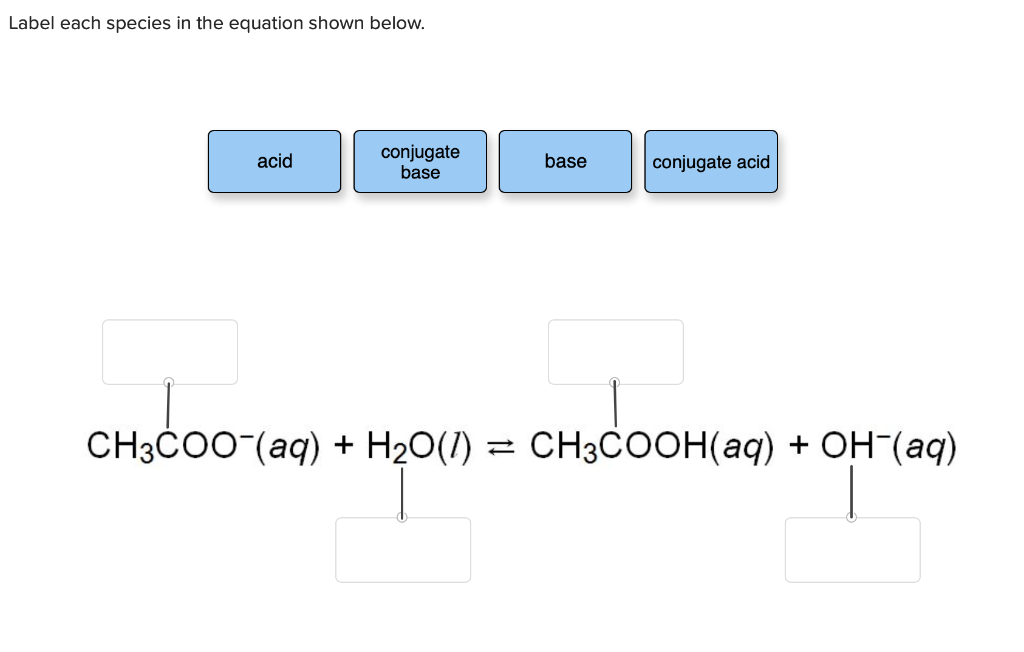
Solved Label each species in the equation shown below. acid
How does one find the conjugate acids and bases in a chemical reaction? - Quora
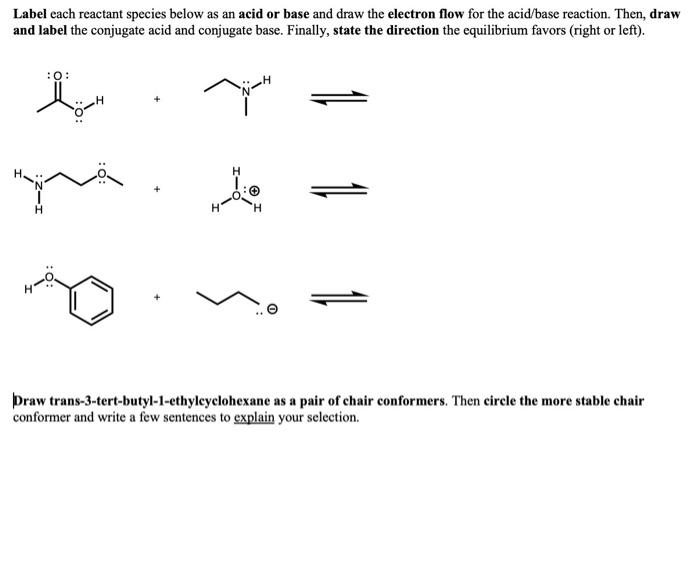
Solved Label each reactant species below as an acid or base

Organic Acids and Bases Practice Problems - Chemistry Steps
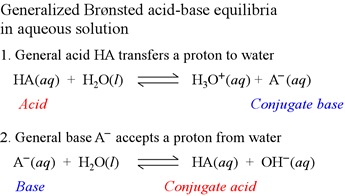
Acids and bases: The Brønsted-Lowry definition
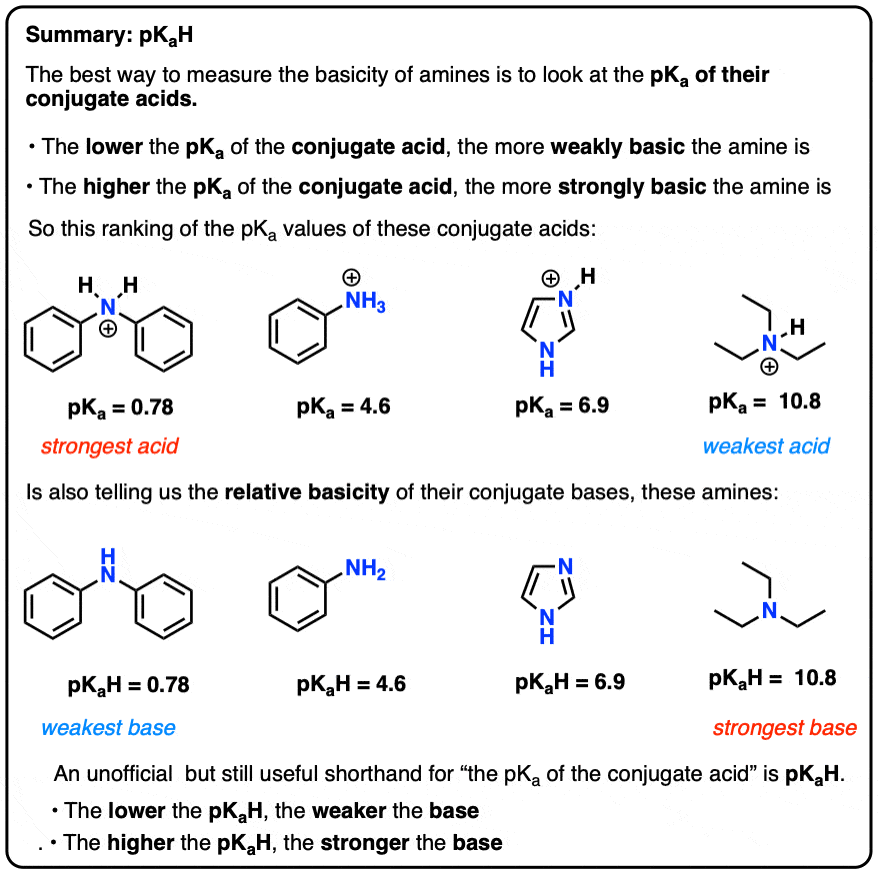
Amine Basicity Is Measued By The pKa Of Its Conjugate Acid (pKaH)

4.8 Introduction To Acid-Base Reactions Student, PDF, Acid

Solved For the Brønsted acid-base reaction shown below

Solved (4 + 6 = 10 points) Q22. (A) Write the formula for

Ammonia appears in [TABLE 2-2 ] as both an acid and a conjugate b