
Find the isothermal compressibility `x` of a Van der Walls gas as
4.9 (602) In stock

4.9 (602) In stock

The PV vs 1/V plot for one mole of a van der Waals gas when b=0 and T=300 K is shown below. The value of the van der Waals constant a (units

Find the isothermal compressibility `x` of a Van der Walls gas as a function of volume
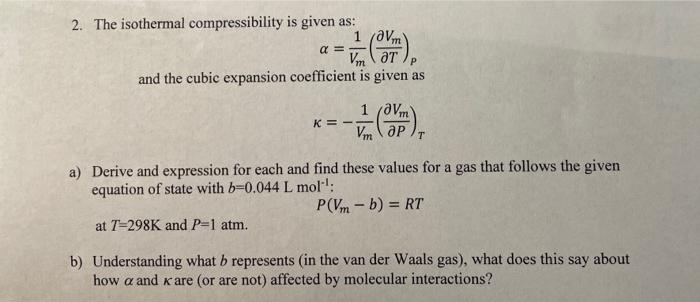
Solved 2. The isothermal compressibility is given as

Answered: Quantities such as the isothermal…

Van der Waals equation - Wikipedia
Find the value of co-efficient of volume expansion β and isothermal compressibility K for a Van der Waals' gas obeying - Sarthaks eConnect

SOLVED: (i) In order to explore the properties of a van der Waals gas near the critical point, introduce dimensionless variables: T = 1 + t, P = 1 + p, V =

Solved I. Coefficient of thermal expansion β and isothermal

Van der Waals Equation:Calculate the expansion and compresibility coefficient