
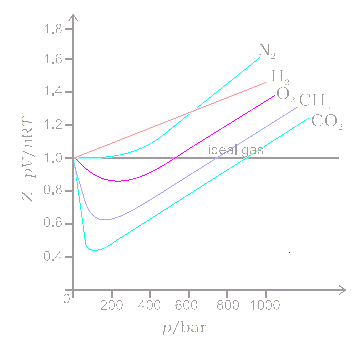
At a high pressure, the compressibility factor (Z) of a real gas is us
4.5 (71) In stock

4.5 (71) In stock
At high P. P gt gt (n^(2)a)/(V^(2)) So ‘a’ can be neglected.

Gas Z Factor Calculator: Dranchuk-Abou-Kassem · PVT Solver
Van der waals equation: Derivation, Explanation
The compressibility factor for a real gas at high pressure is (a) 1+RT/pb (b) 1 (c) 1+pb/RT (d) 1-pb/RT - Sarthaks eConnect

3.3: Real gas and compressibility factor - Engineering LibreTexts

Compressibility Chart - an overview

The compressiblity factor Z for 1 mole of a real gas at low pressure can be written as

Objectives_template

Ch2, Lesson E, Page 9 - Generalized Compressibility Chart

gas laws - How to find the temperature relationship between the isotherms in a compressibility factor (Z) vs pressure graph? - Chemistry Stack Exchange

The ratio of Van Der Waal's constants a and b, ((a)/(b)) has the dime
At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is

The density of air is 0.001293 gm/ml at S.T.P. It's vapour density is

COMPRESSIBILITY FACTOR
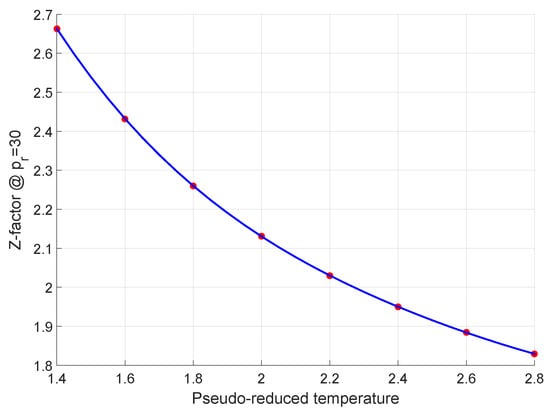
Energies, Free Full-Text

Determine Compressibility of Gases