
What is the compressibility factor (Z) for 0.02 mole of a van der Waal
4.7 (695) In stock

4.7 (695) In stock
(d) (0.1+(1000xx(0.02)^(2))/(V^(2)))V=20xx0.02 =0.1V^(2)-0.4V+0.4=0 =V^(2)-4V+4=0 implies" "V=2L Z=(PV)/(nRT)=(0.1xx2)/(20xx0.02)=0.5

al Gases f.a What is the compressibility factor (Z) 0.02 mole of a van der Waals' gas pressure of 0.1 atm. Assume the size of gas molecules is negligible. Given: RT =

What is the compressibility factor (Z) for 0.02 mole of a van der Waal

Solved Use the Van der Waals EOS to plot the compressibility

Experimental observations of the effects of intermolecular Van der
58.7 Maximum mass of hydrogen is present in(1) 0.1 mol of CH1206(2) 1.5 mol of NH3(3) 22.4 L of H2S(g) at S.T.PSo(4) 0.5 g molecule of CeH

0.585%NaCl solution at 27∘C has osmotic pressure of

Van Der Waals Equation - an overview
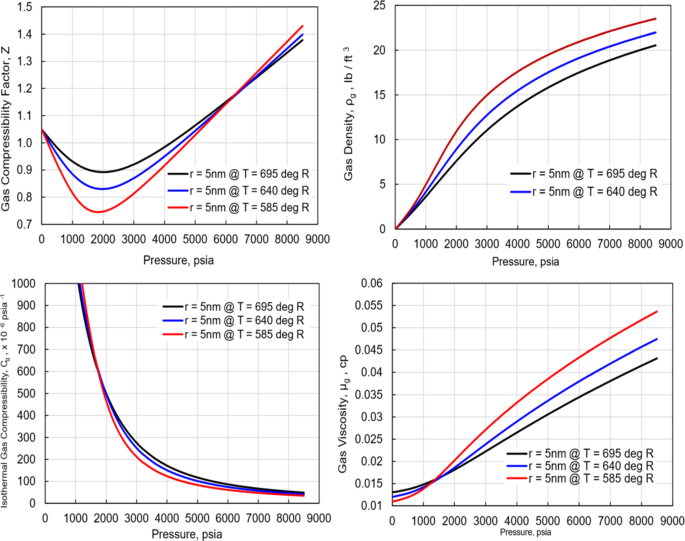
Investigation of the Properties of Hydrocarbon Natural Gases Under

If `Z` is a compressibility factor, van der Waals' equation at low

Van Der Waals Equation - an overview

Real Gases, PDF, Gases

SOLVED: The van der Waals constants for SO2 are a = 6.775 atm L^2

For one mole of a van der Waals gas when b = 0 and T = 300 K , the PV

Compressibility factor (Z) for a van der Waals real gas at critical po