
Which of the following statements is/are correct? (a) all real gases are less compressible
5 (415) In stock

5 (415) In stock
Which of the following statements is/are correct? (a) all real gases are less compressible than ideal gas at high pressures? (6) hydrogen and helium are more co

Role of Mach Number in Compressible Flows
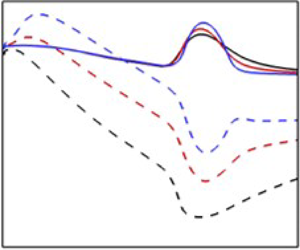
Prandtl number effects on the hydrodynamic stability of compressible boundary layers: flow–thermodynamics interactions, Journal of Fluid Mechanics

Which of the following statement is/are true regarding a real gas? Boyle temperature is the temperature which the slop of the curve between la) Boyle tem compressibility factor vs P is same

Van der Waals Equation, Definition & Examples - Lesson

Volume and Mass Flow Calculations for Gases

Which of the following statements is/are correct? (a) all real gases are less compressible

Van der Waals Equation - Derivation, Formula, Units - Chemistry
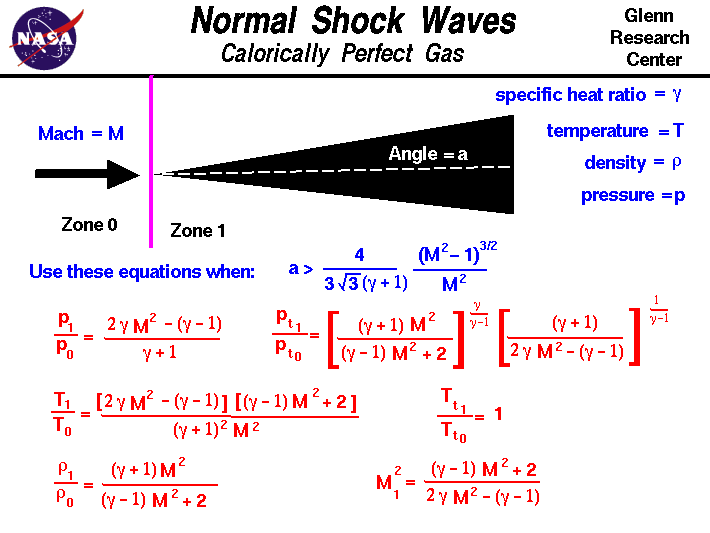
Normal Shock Wave Equations

REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR
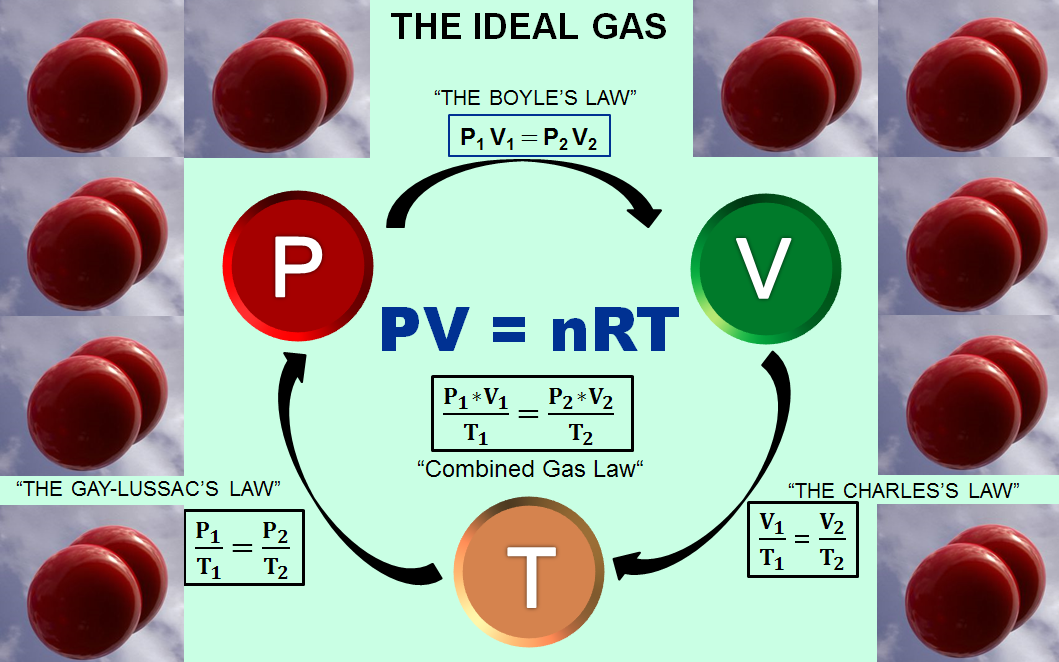
THE 3rd STATE OF MATTER – What is an Ideal Gas? – Computer Aided Design & The 118 Elements
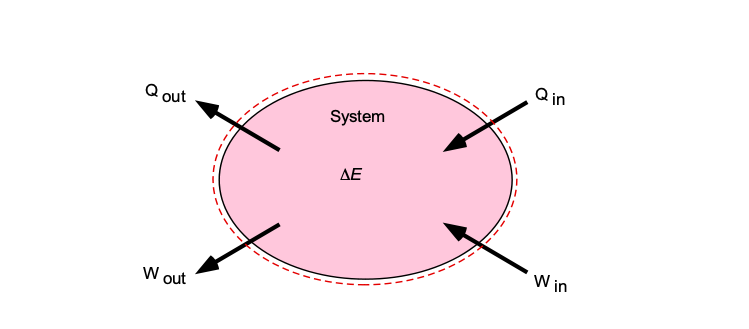
Energy Equation & Bernoulli's Equation – Introduction to Aerospace Flight Vehicles

SOLVED: Make judgment of the following statements and justify your

Difference Between Solid, Liquid, and Gas In Tabular Form

When Z gt 1, real gases are difficult to compress

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?