
What is the compressibility factor (Z) for 0.02 mole of a van der
4.7 (646) In stock

4.7 (646) In stock

An ideal gas at a given state expands to a fixed final volume first at constant pressure and then at constant pressure and then at constant temperature. How is work calculated for

Consider the equation Z=P V/R T. Which of the following statements is correct? (a) When Z1, real

What is the compressibility factor (Z) for 0.02 mole of a van der Waal
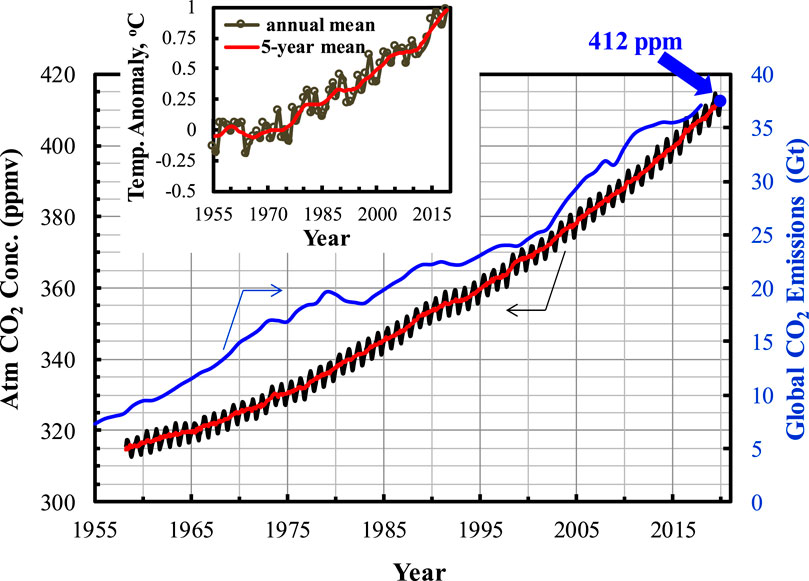
Frontiers Carbon Capture From Flue Gas and the Atmosphere: A Perspective

For a van der Waal's gas, determine Boyle Temperature (given a = 4.5 a

jo 22] What is the compressibility factor (Z) 0.02 mole of a van der Waals' gas pressure of 0.1 atm. Assume the size of gas molecules is negligible.

Solved Please answer all the questions and explain how the

Reascon (R) Even at low pressures, repulsive forces dominate in hydrogen ..

58.7 Maximum mass of hydrogen is present in(1) 0.1 mol of CH1206(2) 1.5 mol of NH3(3) 22.4 L of H2S(g) at S.T.PSo(4) 0.5 g molecule of CeH

Van Der Waals Equation - an overview

Development of Graphical Methods for Estimating the Diffusivity Coefficient of Gases in Bitumen from Pressure-Decay Data

What is the compressibility factor (Z) for 0.02 mole of a van der Waals' gas at pressure of 0.1 a
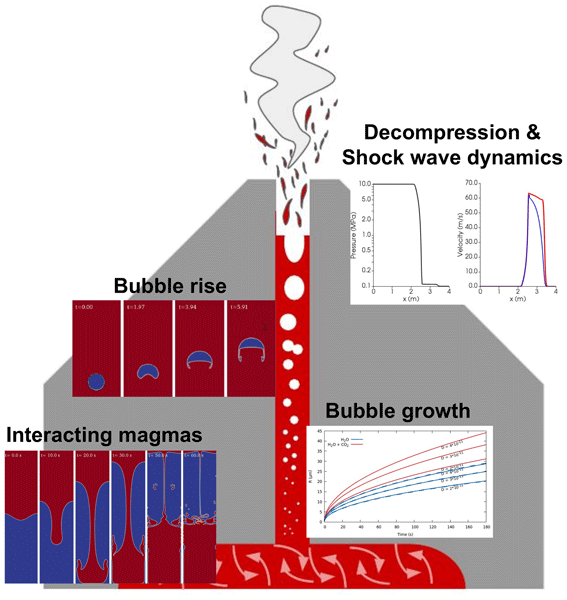
GMD - MagmaFOAM-1.0: a modular framework for the simulation of magmatic systems

Real Gases, PDF, Gases