
UNUB At Boyle temperature, the value of compressi factor Z has a
4.5 (528) In stock

4.5 (528) In stock
Click here:point_up_2:to get an answer to your question :writing_hand:unubat boyle temperature the value of compressifactor z has a value of one over a
Click here👆to get an answer to your question ✍️ UNUB At Boyle temperature- the value of compressi factor Z has a value of one over a wide range of pressure- This is due to the fact that in the van der Waals equation -1- The constant a is negligible and not b -2- The constant b is negligible and not a -3- Both the constant a and b are negligible -4- Attraction balances repulsion

Deviation From Ideal Gas Behavior - Study Material for IIT JEE

68. The compressibility factor (z) an ideal gas is equal to which of the following values? (A) Zero (B) Less than one (C) Equal to one

PDF) AS VT Visual Touch 2.22.2024Alternative Sight Vision Transducer Visual Touch (AS VT2) Ongoing Research compilation1 portends Blindness Breakthru

The compressibility factor for a real gas at high pressure is

PDF) Effect of Temperature and Z-Factor on Casing Designusing Kick

Chemistry_1 - Flipbook by NOWFIYA N

1.7: Connecting the van der Waals and the viral equations- the Boyle temperature - Chemistry LibreTexts
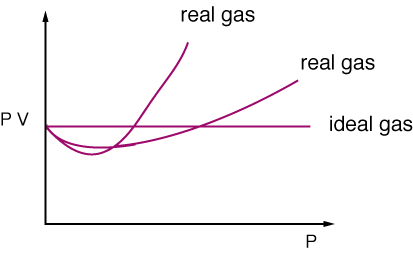
Deviation From Ideal Gas Behavior - Study Material for IIT JEE

Txt.04 - Std'11 - Chemistry - Part-I by Saurabh Suman - Issuu

PHYSICS NOTES. O' LEVEL(S1-S4)

Deviation From Ideal Gas Behavior - Study Material for IIT JEE