
Solved The virial expansion of the compression factor (Z)
4.6 (278) In stock

4.6 (278) In stock

1.5 Real Gases and the Virial Equation - Mail
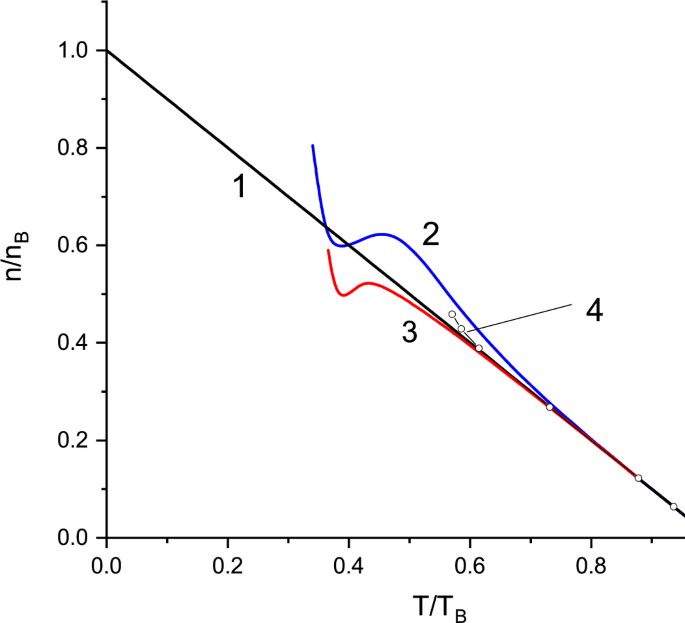
Modified Virial Expansion and the Equation of State

Solved (b) The virial equation of state for a gas, including

The virial form of van der Waal's gas equation is PV=RTleft(1+

SOLVED: The Dieterici equation is another two-parameter equation of state for a one-component gas (much like the van der Waals equation): PV = RT exp(-a/Vm) Question: Write the Dieterici equation for the

Chapter 1. Properties of Gases - ppt download

At 273 K measurements on argon gave B = -21.7 cm$^3$ mol$^{

The virial coefficients predicted by equations of state
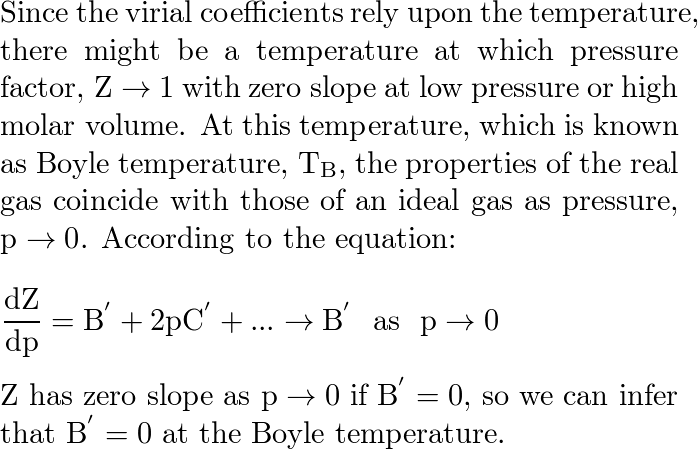
The second virial coefficient of methane an be approximated

Any know how I can relate compressabilty to gibbs free energy so I
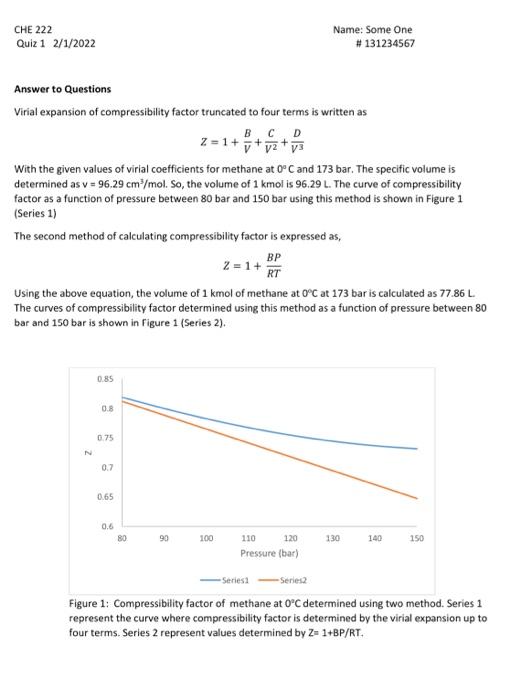
Answer to Questions Virial expansion of

Degrees Conferred by Major ( ) Source: National Center for

⏩SOLVED:At 273 K measurements on argon gave B=-21.7 cm^3 mol^-1