
Microbiological Media Management - SOP & Guideline - Pharma Beginners
4.5 (338) In stock

4.5 (338) In stock
Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.

EU GMP Annex 1

SOP On Procedure For Microbiological Monitoring of Purified Water in Pharmaceutical Company, PDF, Quality Assurance

Stability testing for pharmaceutical products by GMP SOP - Issuu

BIG Benefits: SOPs In The Pharmaceutical Industry

SOP for Isolation and Identification of Microorganisms - Pharma Beginners
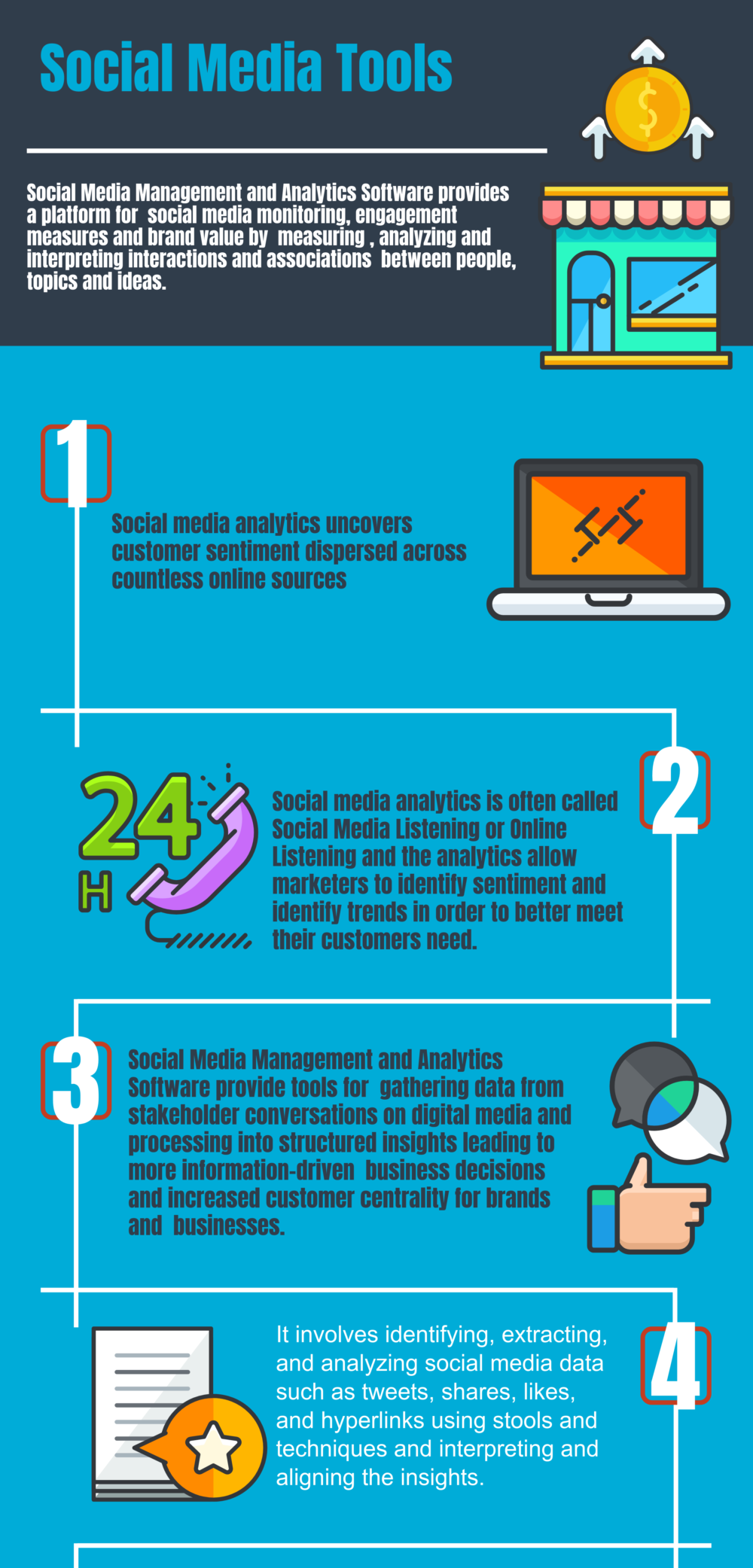
Top 33 Free Social Media Management, Social Media Analytics and Social Publishing Software in 2024 - Reviews, Features, Pricing, Comparison - PAT RESEARCH: B2B Reviews, Buying Guides & Best Practices

Culture media

Qualifying your cleanroom

MICRO 4 SOP For Microbial Monitoring in Drain Point of Pharmaceutical Manufacturing Sites

SOP for Quality Management System (Documentation) in Pharmaceutical Industry

The Use of Microbiological Culture Media Article, PDF
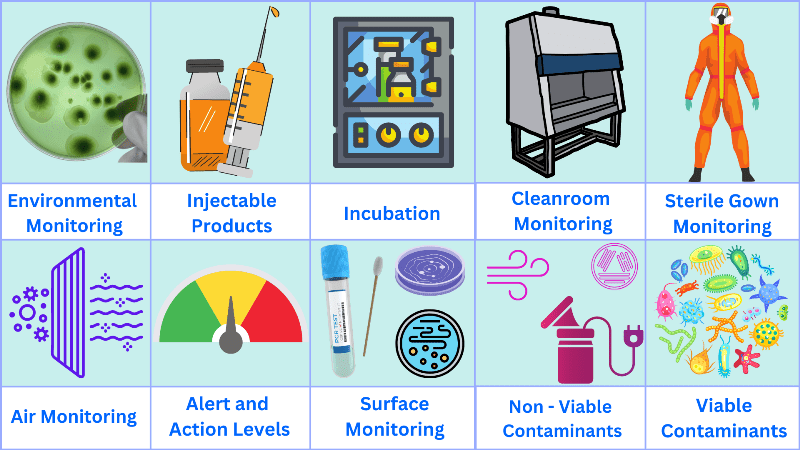
What is environmental monitoring in pharmaceutical industry

Organoids, Free Full-Text