Sacituzumab Earns Regular FDA Approval for TNBC - NCI
4.6 (607) In stock

4.6 (607) In stock
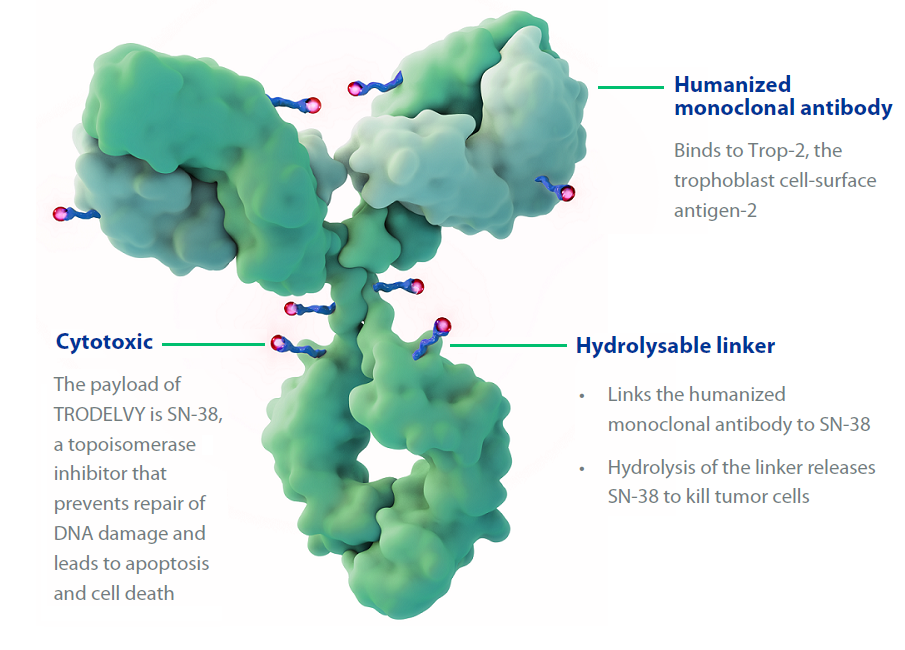
Sacituzumab govitecan (Trodelvy) now has regular FDA approval for people with locally advanced or metastatic triple-negative breast cancer (TNBC), including those with brain metastases. The update follows last year’s accelerated approval of the drug for people with TNBC.
Cureus Advancements in the Treatment of Triple-Negative Breast

Sacituzumab Earns Regular FDA Approval For TNBC NCI
Sacituzumab Govitecan Moves to Second-Line Therapy for Metastatic

Sacituzumab Earns Regular FDA Approval for TNBC - NCI

Sacituzumab Govitecan Moves to Second-Line Therapy for Metastatic
Sacituzumab Earns Regular FDA Approval For TNBC NCI
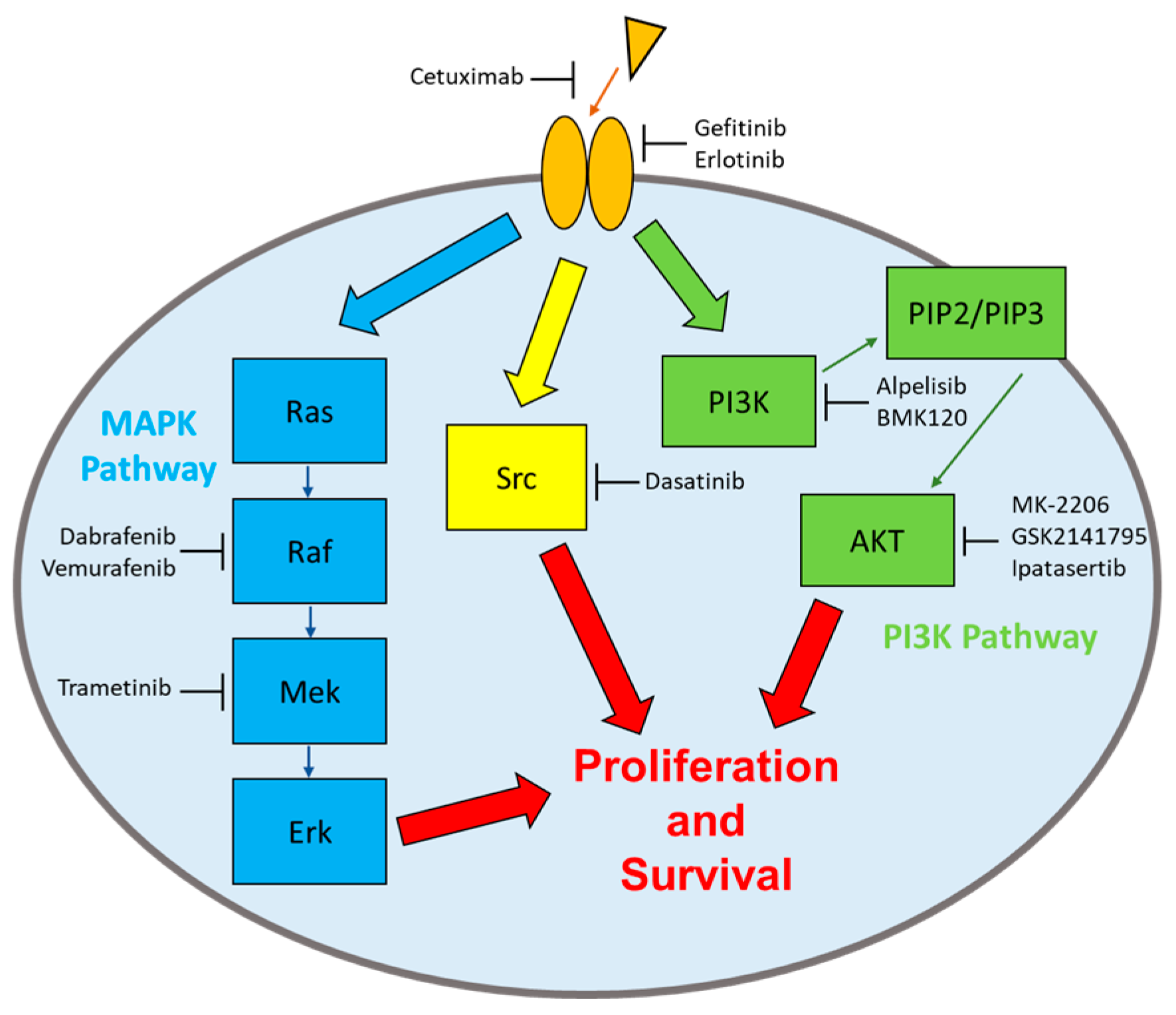
Biomolecules, Free Full-Text
FDA Approvals - Cancer Currents Blog - NCI
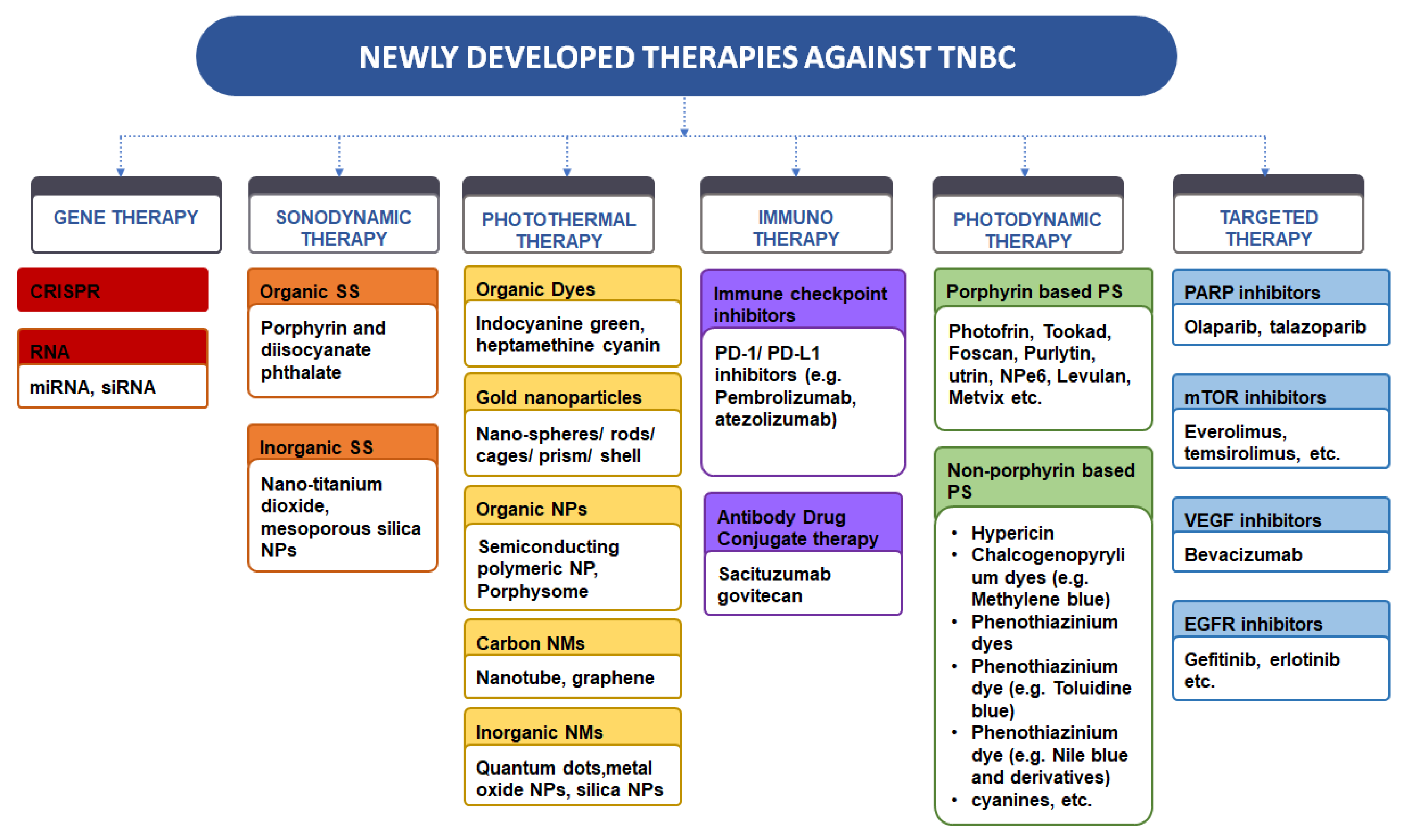
Pharmaceutics, Free Full-Text

Mission Mountain Wilderness

Sacituzumab govitecan in previously treated hormone receptor
Targeting Triple-negative Breast Cancer
Trodelvy for the Treatment of Advanced Triple-Negative Breast Cancer

Recent advances in targeted strategies for triple-negative breast cancer, Journal of Hematology & Oncology

Trodelvy Any Chance Against Dato-DXd and Enhertu.pdf