
Crucial Steps for Singapore Medical Device Registration & HSA Approval
4.9 (535) In stock

4.9 (535) In stock
Discover the crucial steps for successful Singapore medical device registration and HSA approval. Operon Strategist offers expert guidance, classification insights, and comprehensive support. Contact Operon Strategist to learn more and navigate the regulatory landscape with confidence.

Singapore's HSA - Global Regulatory Partners, Inc.

New Requirements for Software Applications in Singapore
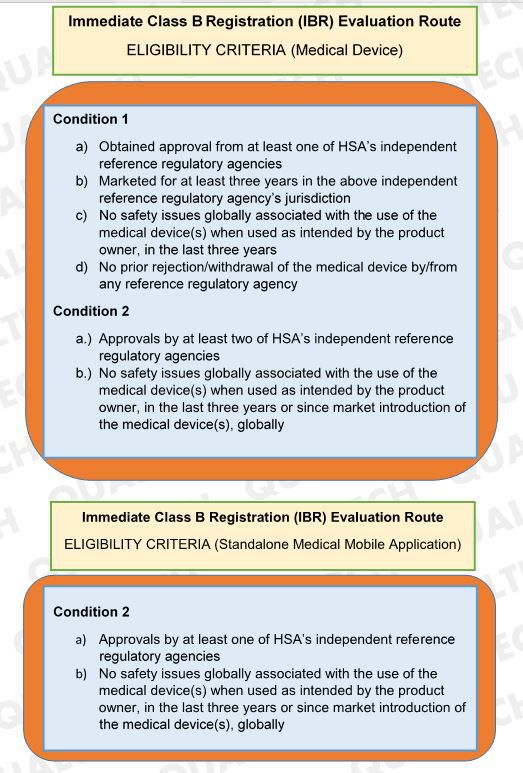
SINGAPORE: HSA Improves Criteria For Immediate Class B Registration (IBR) Evaluation Route – April, 2019
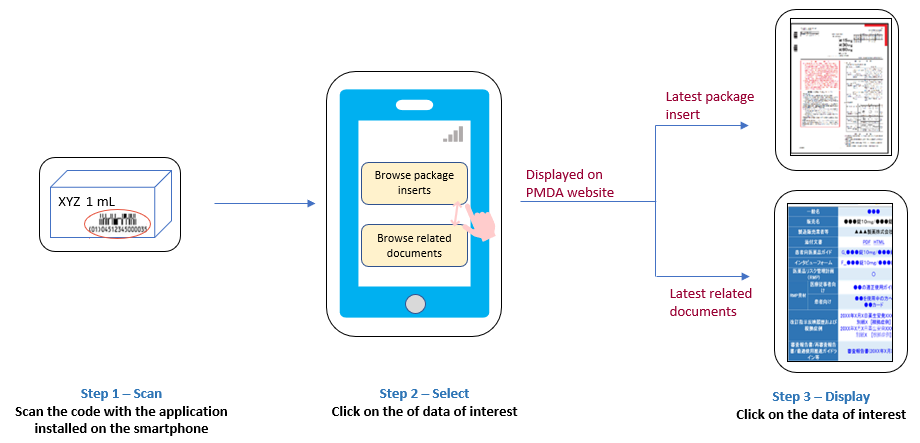
E-labeling and digital transformation in healthcare

Crucial Steps for Singapore Medical Device Registration & HSA Approval

PDF) REGISTRATION AND APPROVAL PROCESS OF MEDICAL DEVICES IN INDIA AND SINGAPORE QR code

Start a Pharmaceutical Company in Singapore in 2023: Steps & License Requirements

HSA IVD Medical Device Pre-Market Approval Registration Service - Clarity Screening

Credevo

on

Understanding the Medical Device Registration Process in Singapore

EXCLUSIVE! Revolutionising Healthcare: Unlocking the Future with Technology – OpenGov Asia
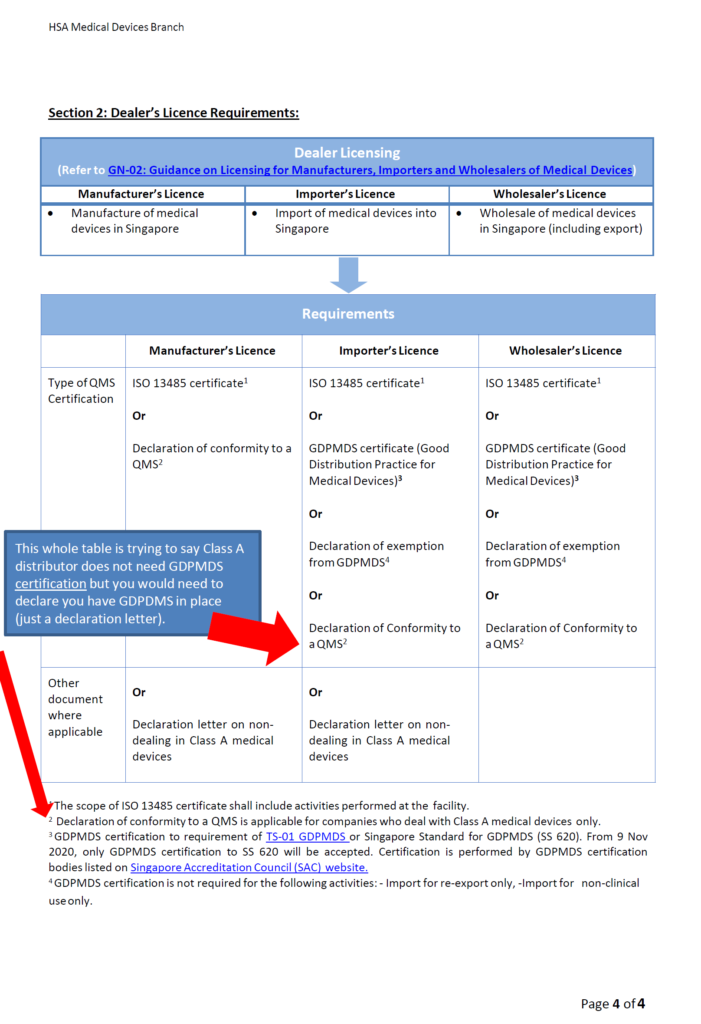
Medical Device registration in Singapore, Health Sciences Authority
Costa Rica regulatory process for medical devices

Kallol Sen on LinkedIn: #medicaldevices #invitrodiagnostics #productregistration