
Five Common Mistakes Submitting a Premarket Notification
4.8 (618) In stock

4.8 (618) In stock
How you can avoid the most common errors made when submitting a 510(k), the “premarket notification,” with simple measures
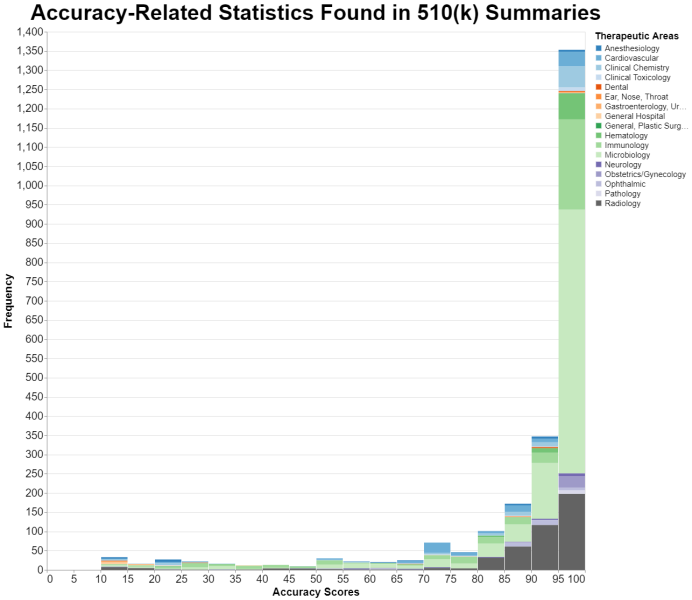
Unpacking Averages: How Accurate Do Class II Medical Devices Need to Be to Obtain 510(k) Clearance?

13 Common Mistakes to Avoid in the 510(k) Submission
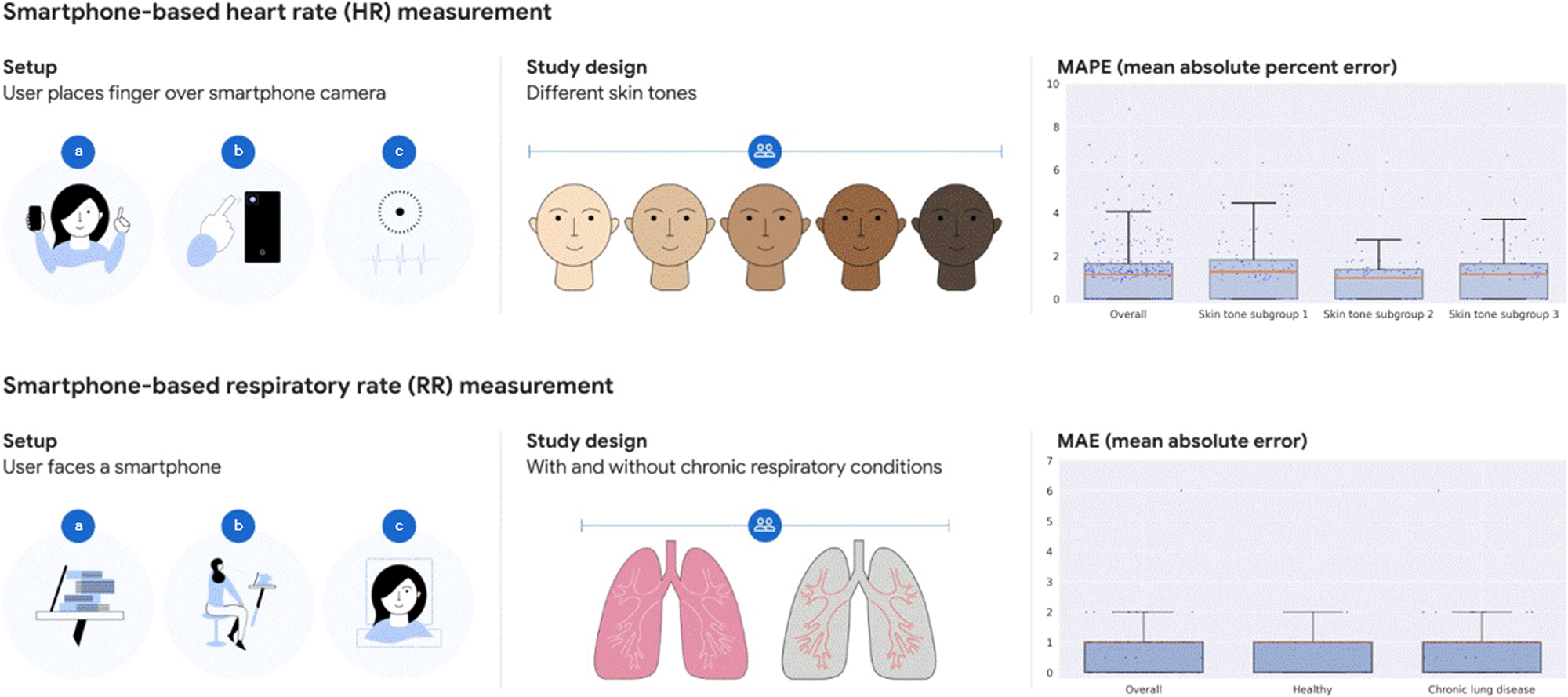
Prospective validation of smartphone-based heart rate and respiratory rate measurement algorithms

Medical Device Academy Blog Archive

Premarket Notification The 510(k) Process

US FDA Pre-Market Notification - 510(k)

Understanding Premarket Trading + 5 Tips You Need To Start
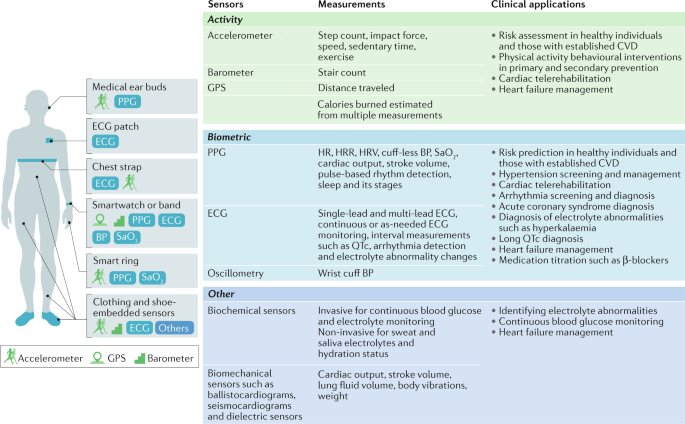
Smart wearable devices in cardiovascular care: where we are and how to move forward
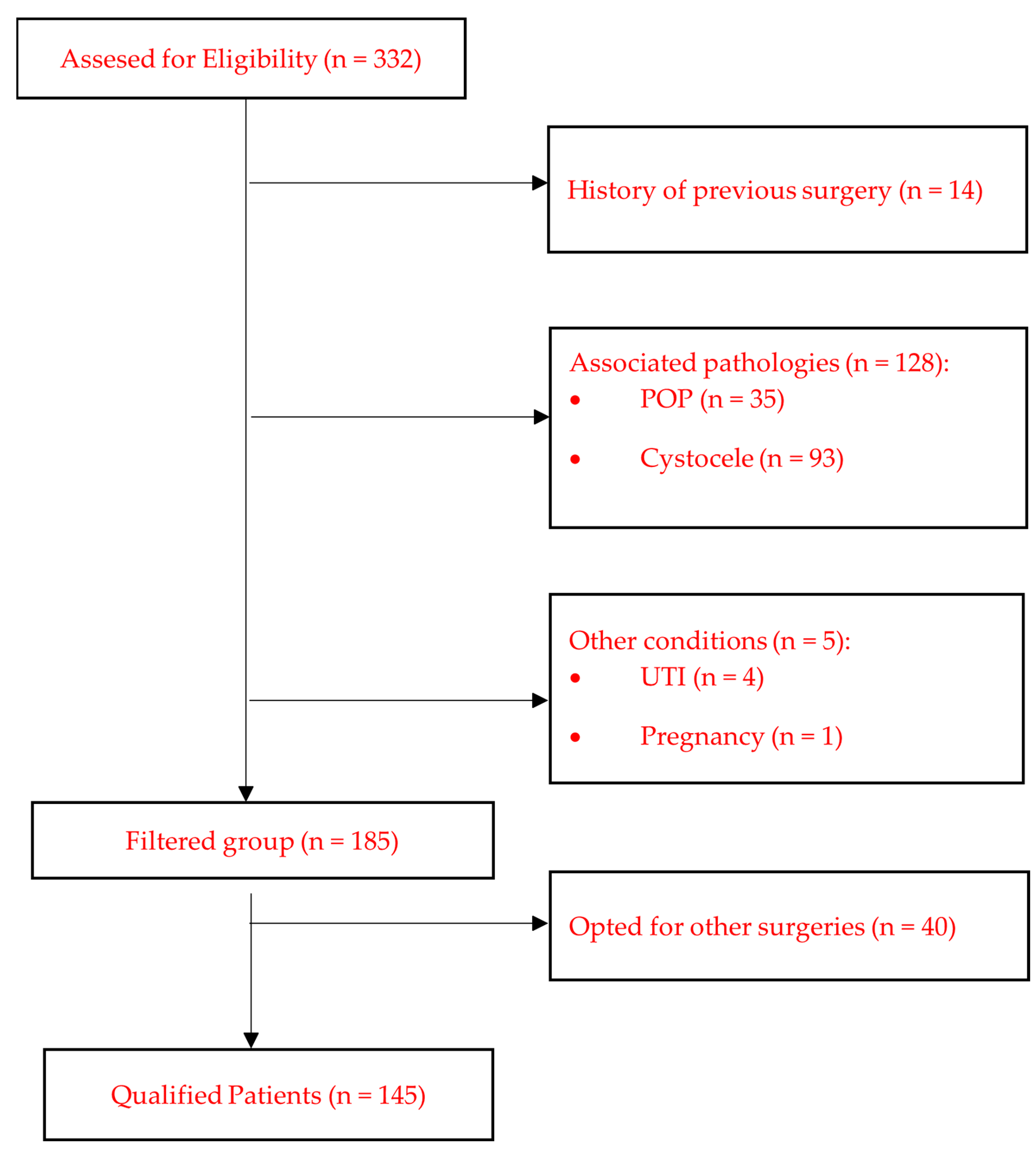
Life, Free Full-Text
:max_bytes(150000):strip_icc()/us-stock-market-time-of-day-tendencies---spy-56a22dc03df78cf77272e6a2.jpg)
Common Intra-Day Stock Market Patterns

US FDA Pre-Market Notification - 510(k)

The FDA 510(k) Process: Setting the Stage for a Successful Submission and Faster Approval – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog