Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
4.9 (795) In stock
4.9 (795) In stock
Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H-H is 0-64 and the bind length of F2 is 1-2- Electronegativities of H and F respectively are 2-1 and 4-1-What is the bond length of HF- 1-0-64 2-0-92 3-0-82 4-0-62
What is an (F2-) bond order? - Quora
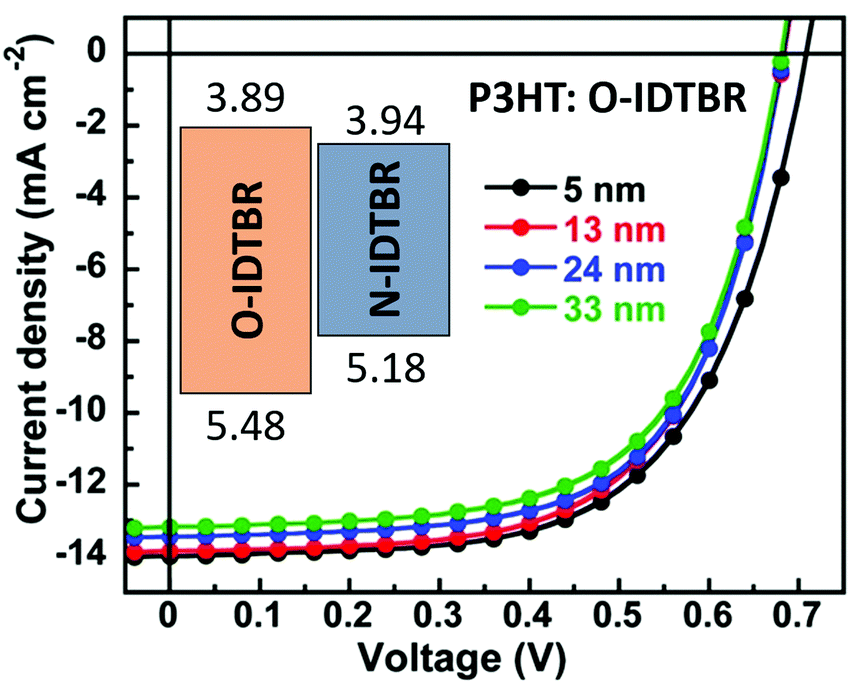
Interlayers for non-fullerene based polymer solar cells: distinctive features and challenges - Energy & Environmental Science (RSC Publishing) DOI:10.1039/D0EE02503H
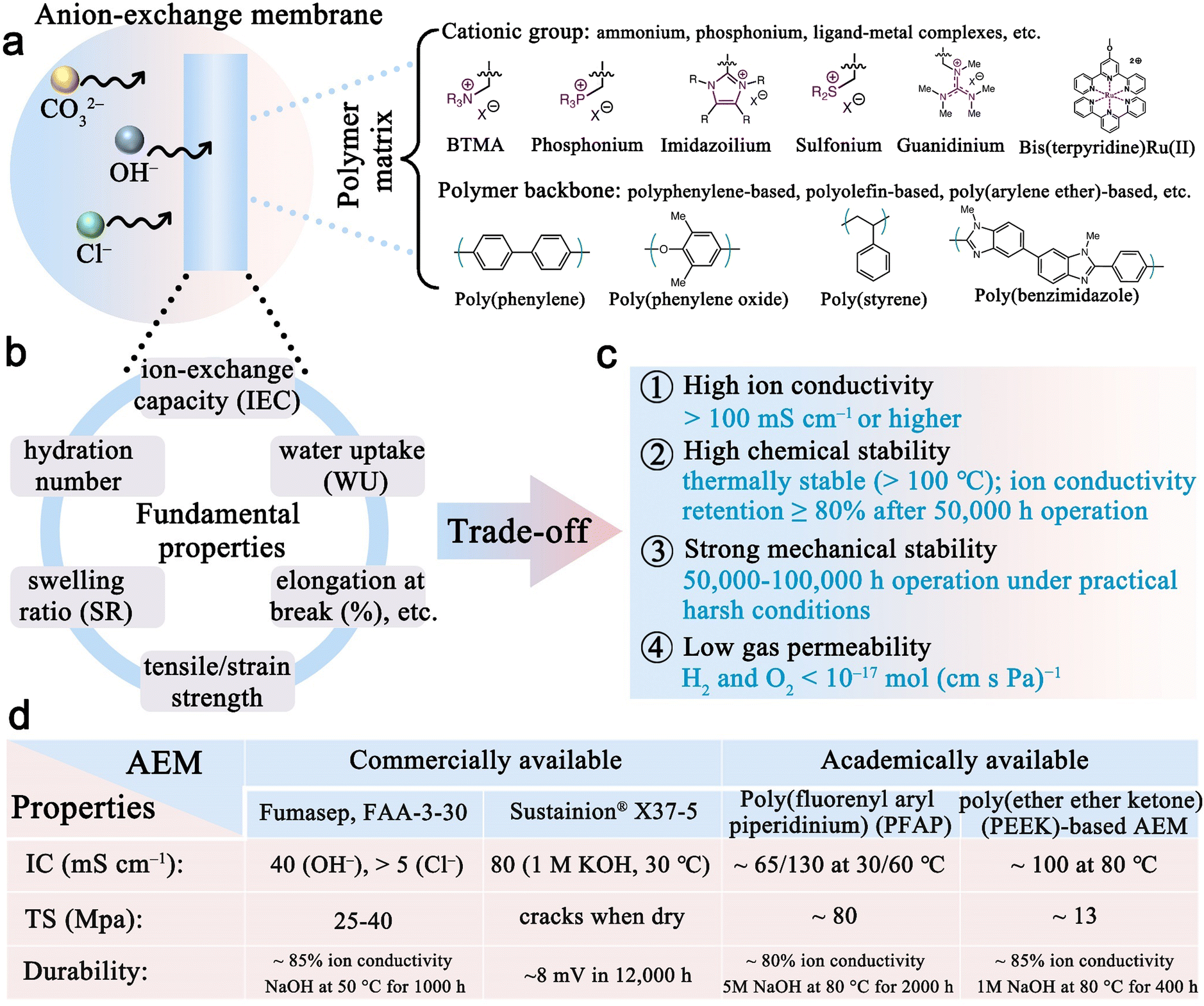
Anion-exchange membrane water electrolyzers and fuel cells - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00038E

6 S.P. - C-4.6 Calculate the theoretical value of bond length in 1. & and are 0.37A and 0.728 respectively. Flectronegativities of P & Hare 20 and 2.1 respectively.
Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
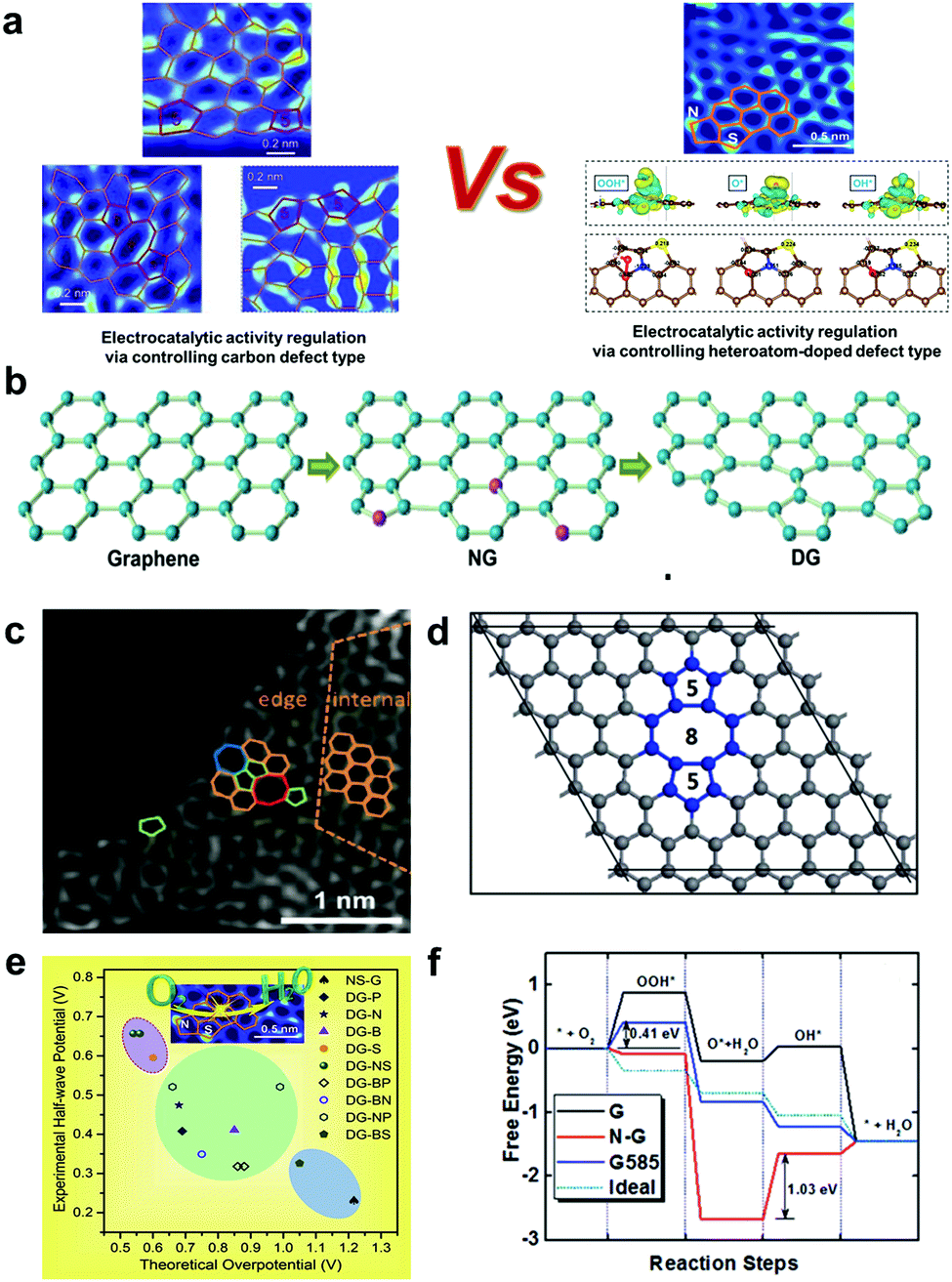
Untangling the respective effects of heteroatom-doped carbon materials in batteries, supercapacitors and the ORR to design high performance materials - Energy & Environmental Science (RSC Publishing) DOI:10.1039/D1EE00166C
In which OO bond is length greater, H2O2 or F2O2? - Quora

Patai S., Rappoport Z. (Eds.) - The Chemistry of Organic Selenium and Tellurium Compounds. v.1, PDF, Functional Group

Chemical Engineering Reviewer Edited, PDF, Molecules

The Chemistry NH2 NO NO2 Related Groups

Ornl 4344 by The E Generation - Issuu