
Bev Shea – OMERACT
4.8 (505) In stock

4.8 (505) In stock

PDF) Looking for important change/differences in studies of responsiveness. OMERACT MCID Working Group

Late-Stage Knee and Hip Osteoarthritis Composite Measure – OMERACT

IFH Associate Faculty - Rutgers
OMERACT consensus-based operational definition of contextual factors in rheumatology clinical trials: A mixed methods study

Updating the Psoriatic Arthritis (PsA) Core Domain Set: A Report from the PsA Workshop at OMERACT 2016. - Abstract - Europe PMC

Updating the Psoriatic Arthritis (PsA) Core Domain Set: A Report from the PsA Workshop at OMERACT 2016. - Abstract - Europe PMC

Safety – OMERACT

Late-Stage Knee and Hip Osteoarthritis Composite Measure – OMERACT

PDF) International Patient and Physician Consensus on a Psoriatic Arthritis Core Outcome Set for Clinical Trials
Towards consensus in defining and handling contextual factors within rheumatology trials: An initial qualitative study from an OMERACT working group
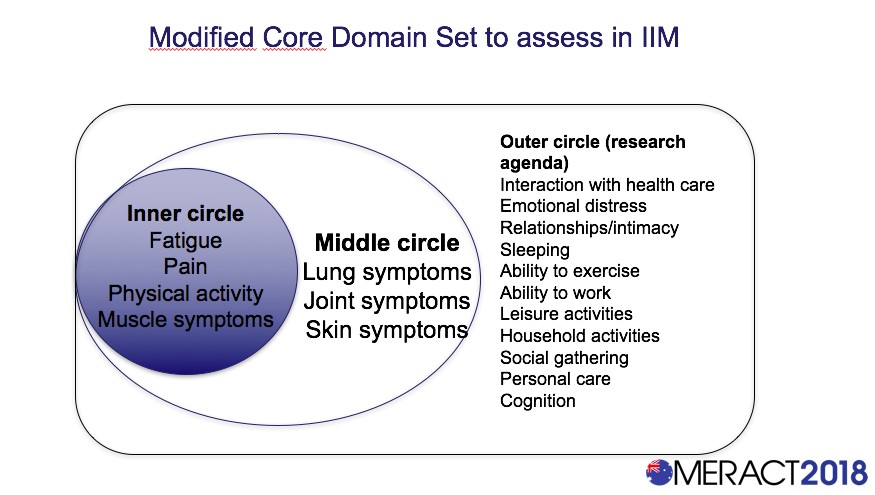
A Draft Modified Core Domain Set for Patient-Reported Outcomes (PRO) in Patients with Idiopathic Inflammatory Myopathies (IIM): An Omeract Report - ACR Meeting Abstracts