200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
5 (330) In stock
5 (330) In stock
200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
200 g of a sample of limestone liberates 66 g of CO2 on heating- The percentage purity of CaCO3 in the limestone is Options-a- 95

Doc 117 b p s xi chemistry iit jee advanced study package 2014 15 by S.Dharmaraj - Issuu

Basic Chemical Calculations-Merged, PDF, Mole (Unit)
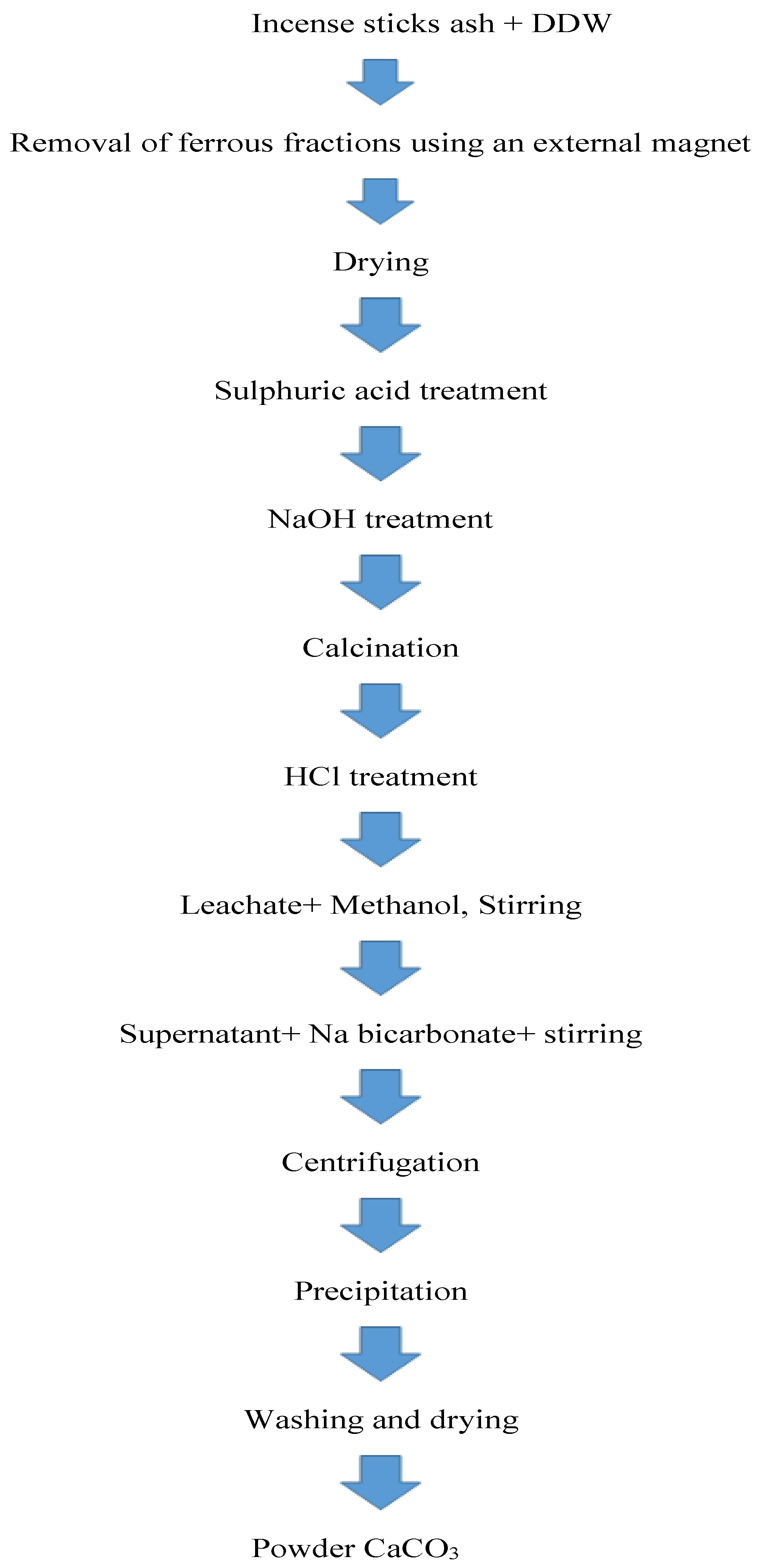
Applied Sciences, Free Full-Text

ESTERFIP, A TRANSESTERIFICATION PROCESS TO PRODUCE
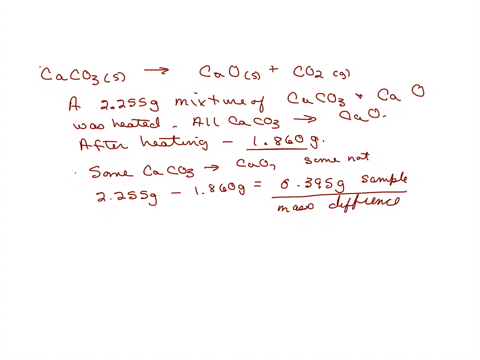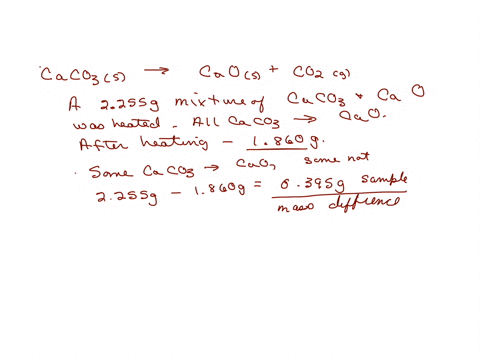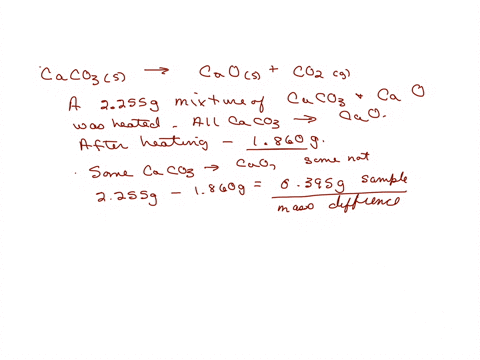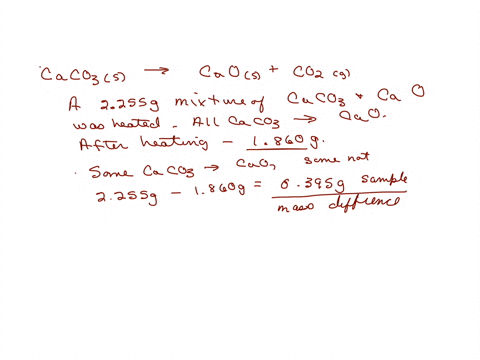
SOLVED: The weight percentage of limestone is as follows: CaCO3 = 93%, MgCO3 = 5%, Insoluble = 2%. This limestone is burned in a vertical kiln to produce lime (CaO, MgO, Insolubles).

Unical Science 2o22
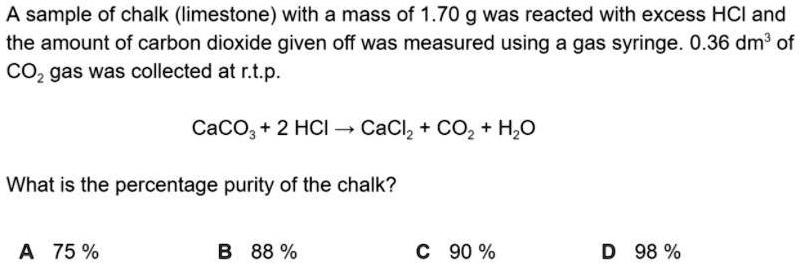
SOLVED: What is the percentage purity of the chalk? A sample of chalk ( limestone) with a mass of 1.70 g was reacted with excess HCl and the amount of carbon dioxide given

Variations of Stable Isotope Ratios in Nature
Energies, Free Full-Text

Carbon Capture and Storage: Most Efficient Technologies for Greenhouse Emissions Abatement

Calculate the weight of lime (CaO) obtained by heating 200 kg of 95% pure lime stone `(CaCO_(3)).`
When a limestone of mass 150g was heated until it decomposed to CaO, only 63g of CaO were obtained. What is the percentage purity of the limestone? - Quora

Adsorbent Materials for Carbon Dioxide Capture from Large Anthropogenic Point Sources - Choi - 2009 - ChemSusChem - Wiley Online Library