32. 80 g of h2 is reacted with 80 g of o2 to form water. find out
4.5 (115) In stock
4.5 (115) In stock
32. 80 g of h2 is reacted with 80 g of o2 to form water. find out the mass of water obtained.which substance is the limiting reagent.
32- 80 g of h2 is reacted with 80 g of o2 to form water- find out the mass of water obtained-which substance is the limiting reagent

2 g H_2 and 1 g O_2 are allowed to react according to following

52. 80 g of H, is reacted with 80 g of O, to form water. Find out the mass of water obtained. Which substance is the limiting reagent?

Hydrogen Oxygen Fuel Cells - an overview
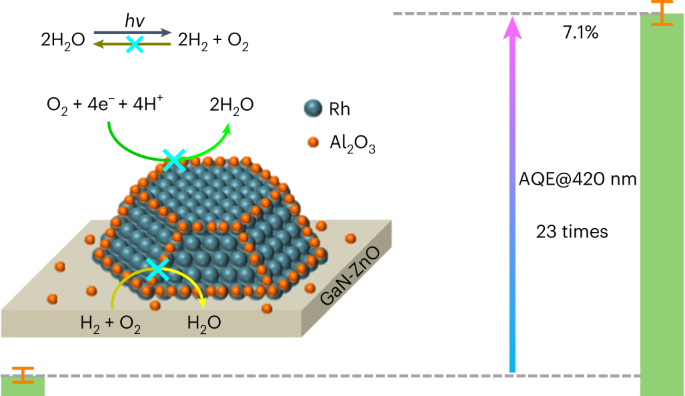
Blocking the reverse reactions of overall water splitting on a Rh

80g of H2 is reacted with 80g of O2 to form water; what are the

20.0 kg of H2 and 32 kg of O2 are reacted to produce H2O.the

Recent Advances in Electrochemical Water Oxidation to Produce

80 Gram h2 is reacted with 80gram o2 to form water find Limiting

Hydrogen and oxygen combine in the ratio of 1:8 by mass to form water

SOLVED: 3.0 g of H2 react with 29.0 g of O2 to form H2O (i) which
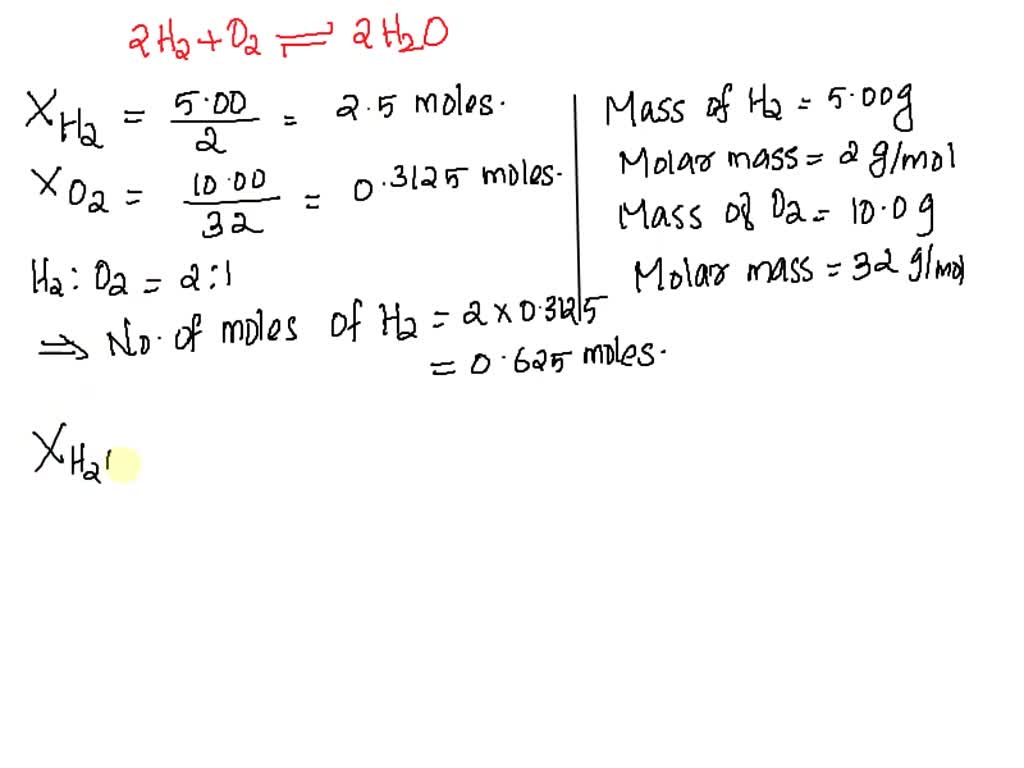
SOLVED: Which is the limiting reactant when 5.00 g of H2 and 10.0