2.t 300 K, 36 g of glucose present per litre in itssolution has an
4.5 (540) In stock
4.5 (540) In stock
2.t 300 K, 36 g of glucose present per litre in itssolution has an osmotic pressure of 4.98 bar. If theosmotic pressure of solution is 1.52 bar at thesame temperature, what would be itsconcentration?(1) 11 gl 1(3) 36 gl 1(2) 22 gL 1(4) 42 gL 1
2-t 300 K- 36 g of glucose present per litre in itssolution has an osmotic pressure of 4-98 bar- If theosmotic pressure of solution is 1-52 bar at thesame temperature- what would be itsconcentration-1- 11 gl-1-3- 36 gl-1-2- 22 gL-1-4- 42 gL-1

NCERT Solutions for Class 12 Chemistry Solutions

NCERT Solutions for Class 12 Chemistry Chapter 2 Solutions
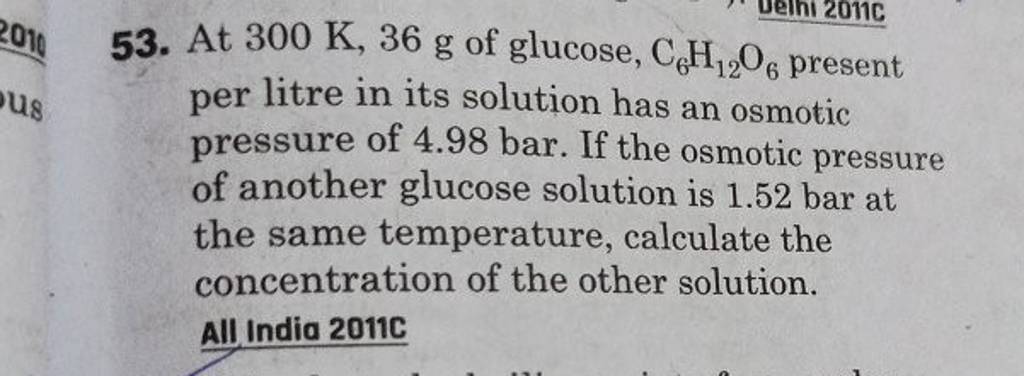
53. At 300 K,36 g of glucose, C6H12O6 present per litre in its solutio..

At 300 K,36 g of glucose present per litre in its solution has an osmotic..

If fa ..+ 3 Oct. GL-4

NCERT Solutions for Class 12 Chemistry Chapter 2 Solutions
CH104: Chapter 7 - Solutions - Chemistry

At 300K, 36g of glucose present in one litre of its solution has an os - askIITians

At 300K, 26g of glucose present in a litre of its solution has an osmotic pressure of 4.98 bar.