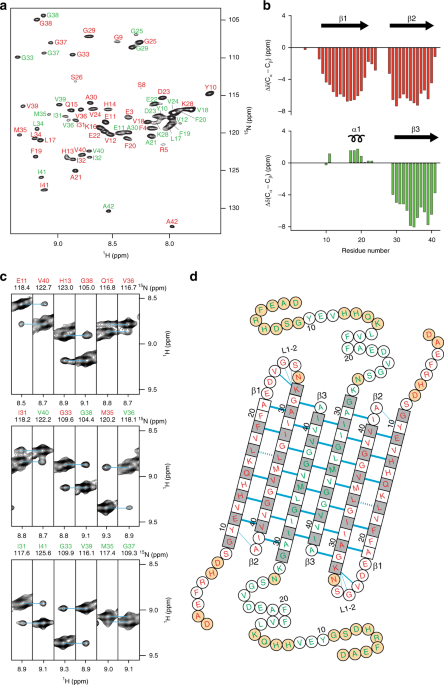
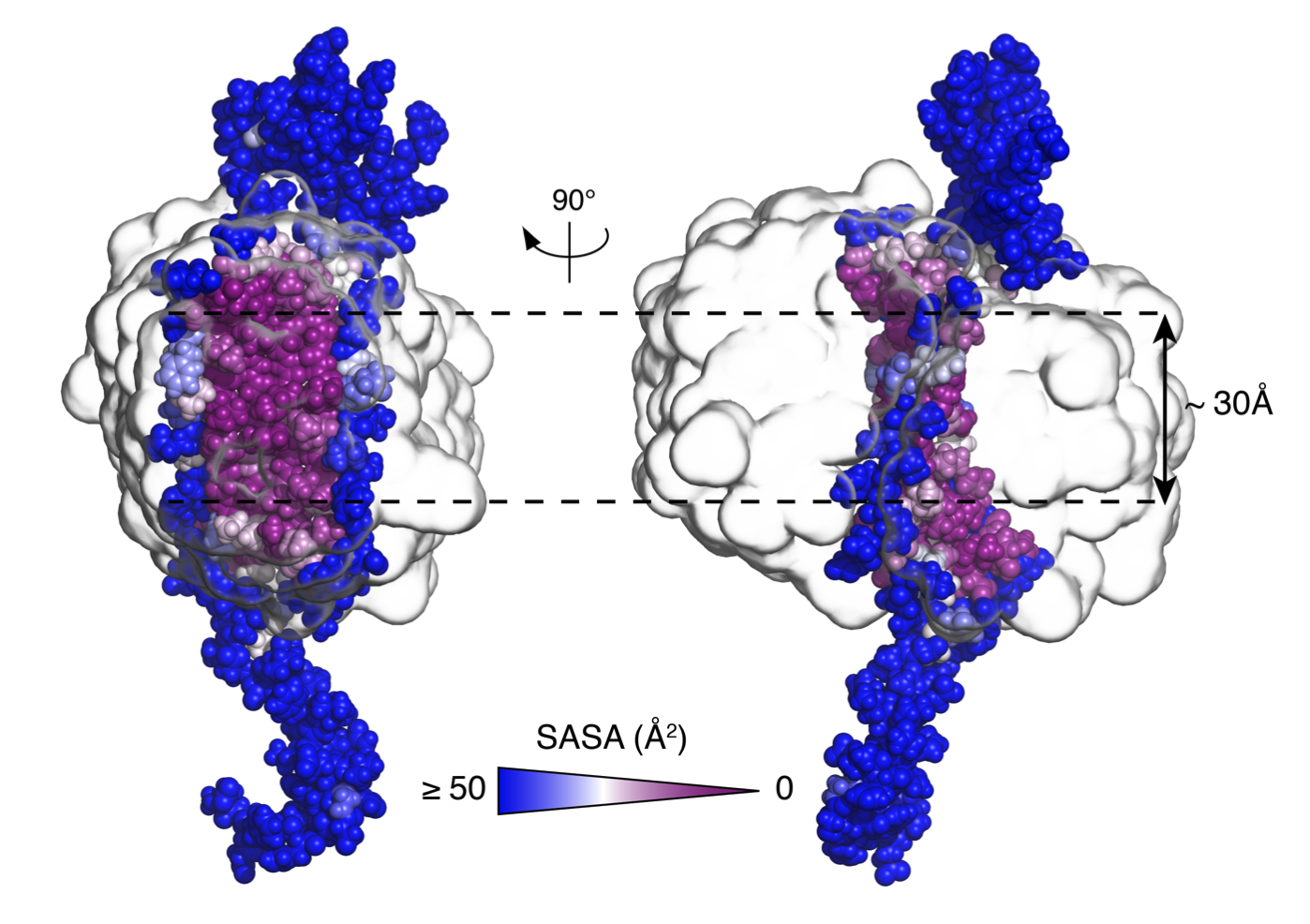
Aβ(1-42) tetramer and octamer structures reveal edge conductivity
4.8 (361) In stock

4.8 (361) In stock

Molecular dynamics simulations reveal the importance of amyloid-beta oligomer β-sheet edge conformations in membrane permeabilization - ScienceDirect

Eduard Puig, Ph.D. - Freelance
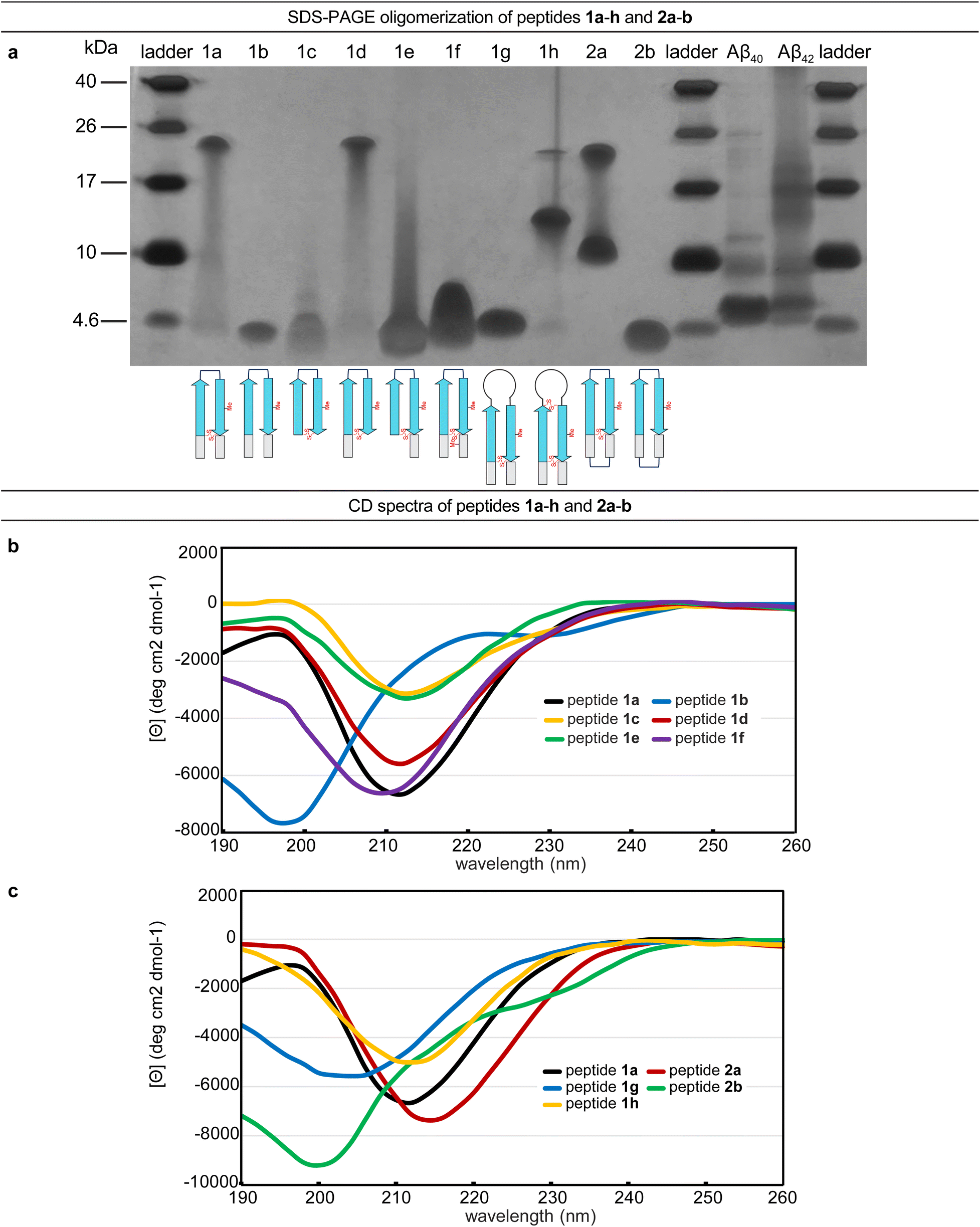
A β-barrel-like tetramer formed by a β-hairpin derived from Aβ - Chemical Science (RSC Publishing) DOI:10.1039/D3SC05185D

Amentoflavone: A Bifunctional Metal Chelator that Controls the

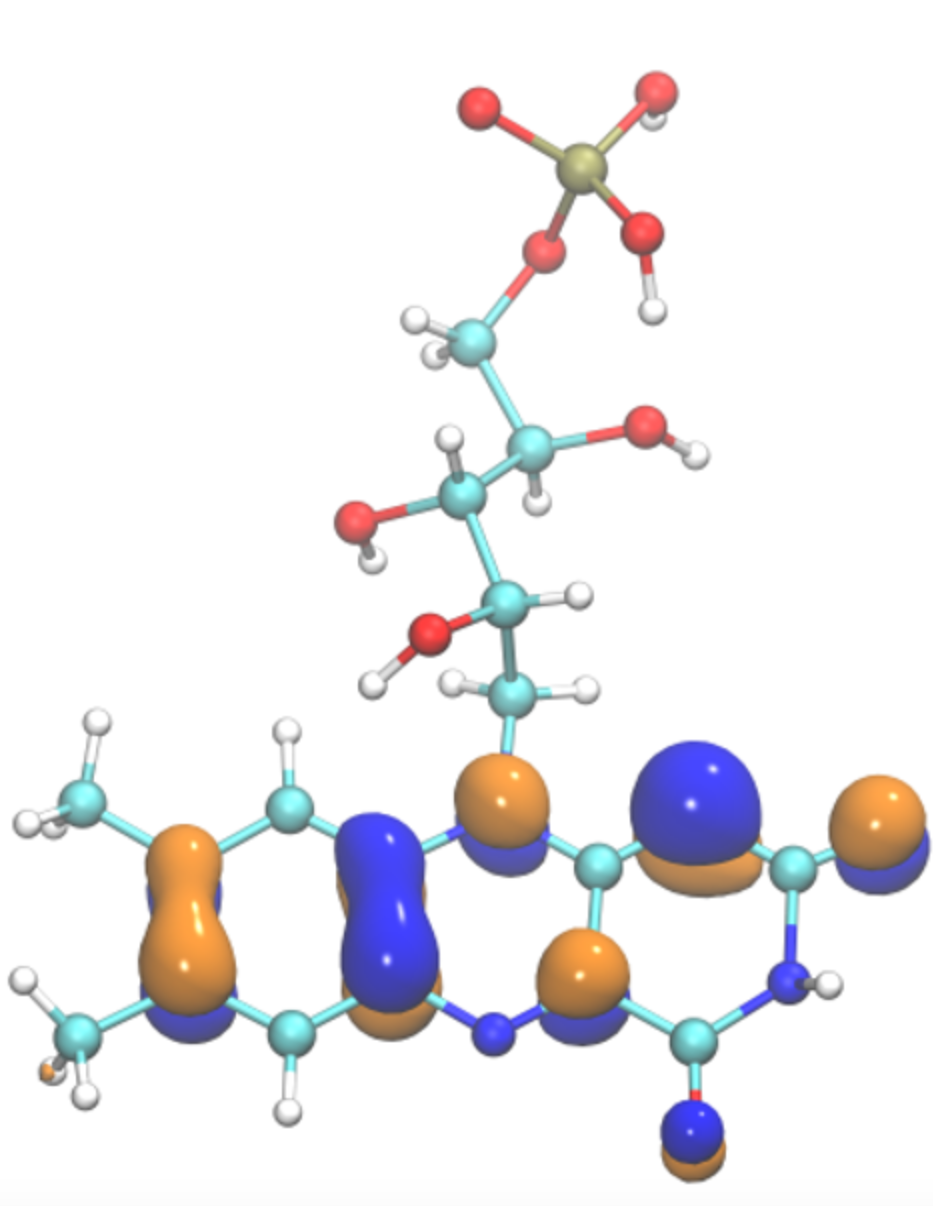
Secondary structure determines electron transport in peptides
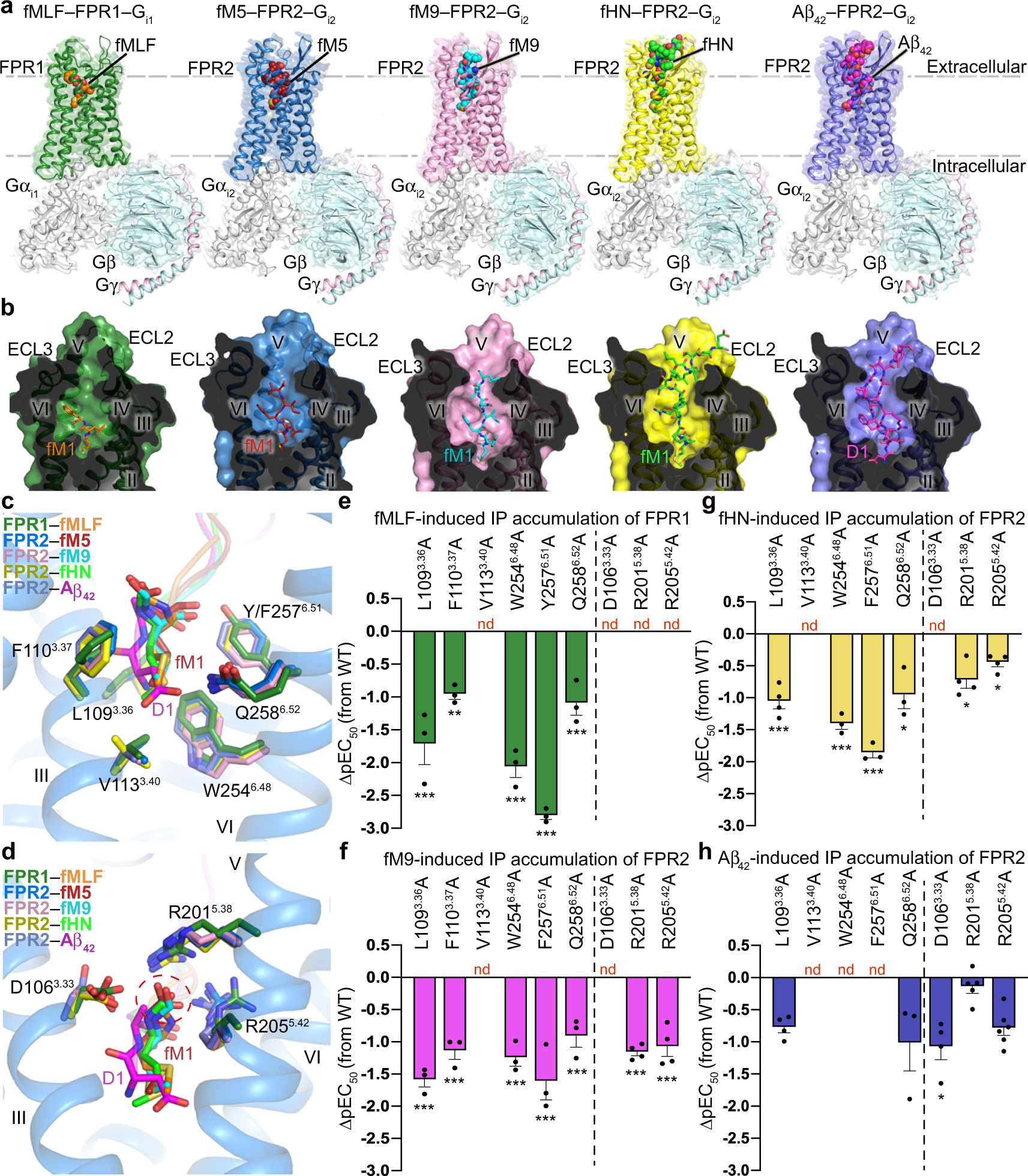
Structural basis of FPR2 in recognition of Aβ42 and
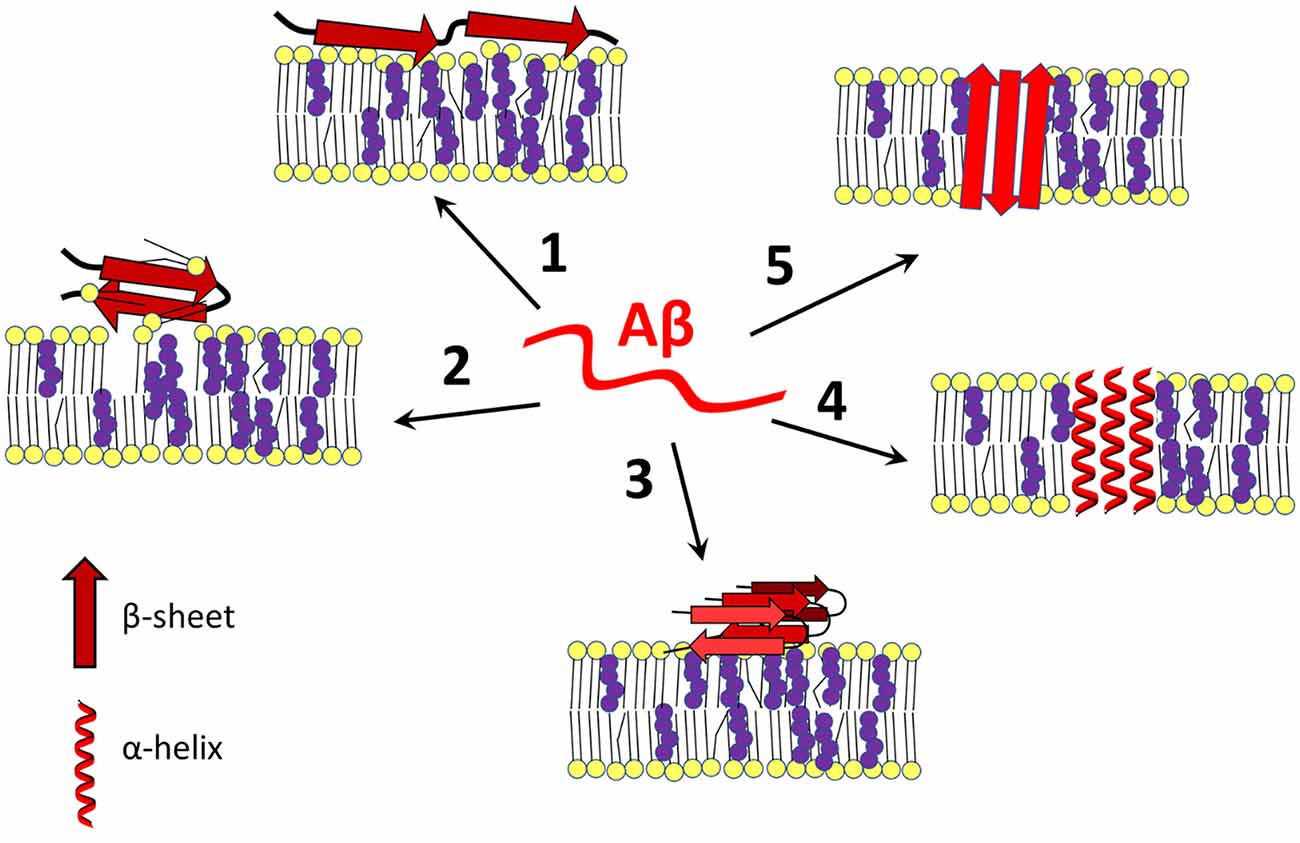
Frontiers Cholesterol as a key player in amyloid β-mediated toxicity in Alzheimer's disease
Molecules, Free Full-Text
Andres Arango

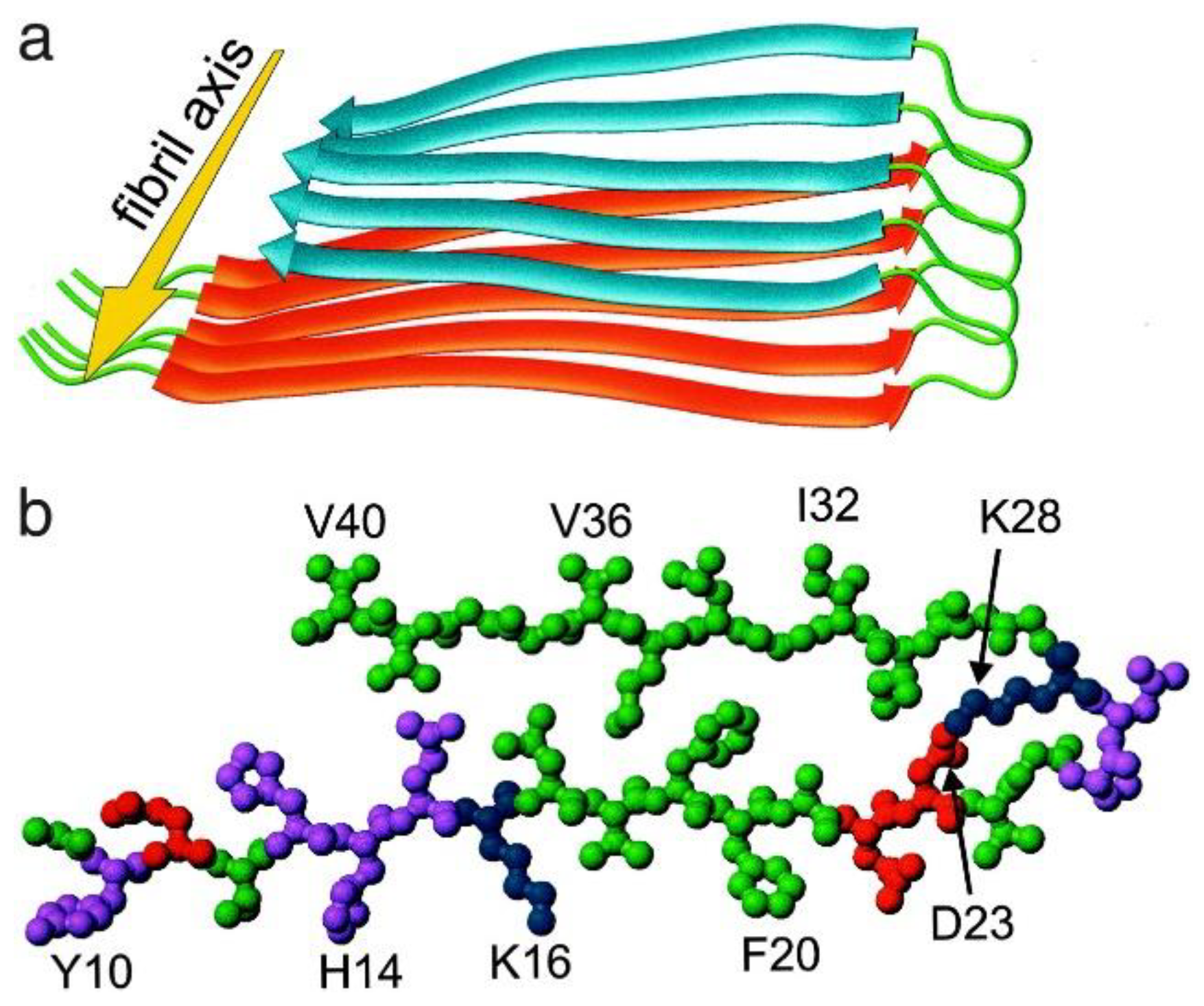
Aβ(1-42) tetramer and octamer structures reveal edge conductivity pores as a mechanism for membrane damage

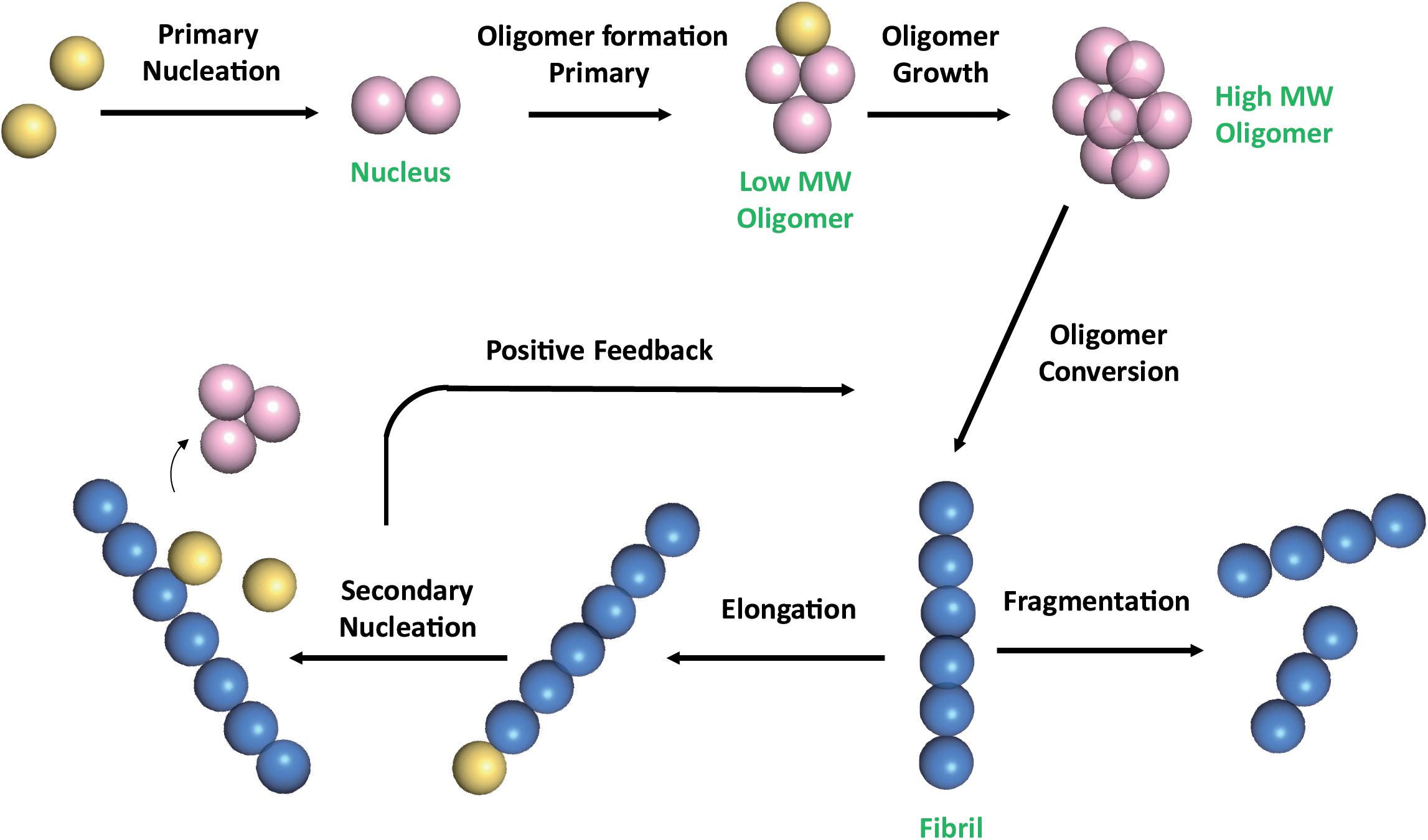
Analyzing Morphological Properties of Early-Stage Toxic Amyloid β Oligomers by Atomic Force Microscopy
Computational Structural Biology and Molecular Biophysics

Molecular dynamics simulations reveal the importance of amyloid-beta oligomer β-sheet edge conformations in membrane permeabilization - ScienceDirect

Single-molecule Mapping of Amyloid-β Oligomer Insertion into Lipid
Frontiers Natural Compounds as Inhibitors of Aβ Peptide Aggregation: Chemical Requirements and Molecular Mechanisms